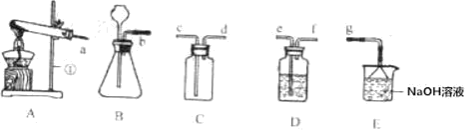��Ŀ����
����Ŀ��ij��ȤС��ͬѧ��ʵ�����Ʊ�������������������̽��ʵ�顣
��1�����������������������Һ�ֽ������Ƿ��й��أ���������¶Ա�ʵ�飺
��3.0g 10%H2O2��Һ��1.0g MnO2���Ȼ�ϣ�
��x g 10%H2O2��Һ��1.0g CuO���Ȼ�ϡ�
����ͬ�¶��£��Ƚ�����ʵ�����O2�Ŀ�����
���з�Ӧ�Ļ�ѧ����ʽ��__________________________��
����x��ֵӦΪ_____________g��
����������������ʱȢ�죬�ɴ˵ó���ʵ�������______________________��
��2����̽����Ӱ�����������Һ�ֽ��ٶȵ�ij�����ء�ʵ�����ݼ�¼���£�
����������Һ������ | ����������Һ��Ũ�� | MnO2������ | ��ͬʱ���ڲ���O2��� | |
�� | 50.0g | 1% | 0.1g | 9mL |
�� | 50.0g | 2% | 0.1g | 16mL |
�� | 50.0g | 4% | 0.1g | 31mL |
��ʵ���У�����O2�����װ����______________�����ţ���
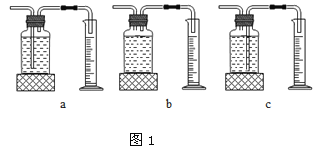
ʵ����ۣ�����ͬ�����£�___________________������������Һ�ֽ��Խ�졣
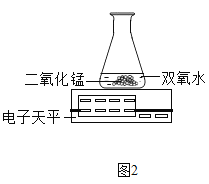
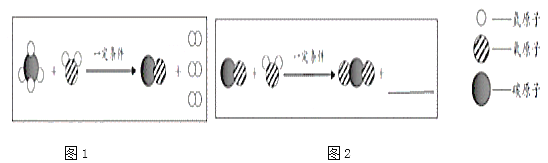
��3��������ͼ2װ�ý���ʵ�飬ͨ���Ƚ���ͬʱ����____________Ҳ�ܴﵽʵ��Ŀ�ġ�
���𰸡� 2H2O2 ![]() 2H2O+O2�� 3.0 ���������������������Һ�ֽ������й� c ����������ҺŨ��Խ�� ��Ӧǰ����ƽ�Ķ���֮��
2H2O+O2�� 3.0 ���������������������Һ�ֽ������й� c ����������ҺŨ��Խ�� ��Ӧǰ����ƽ�Ķ���֮��
����������1�����������ڶ������̵Ĵ������·ֽ�Ϊˮ����������ѧ����ʽΪ��2H2O2 ![]() 2H2O+O2�� �����ƹ���������Һ��Ũ����������ȣ����ܱȽϳ��������̺�����ͭ�Ĵ�Ч�������Ԣ���x����ֵΪ3.0g������������������ʱȢ�죬�ɴ˵ó���ʵ������Ǵ��������������������Һ�ֽ������йأ�
2H2O+O2�� �����ƹ���������Һ��Ũ����������ȣ����ܱȽϳ��������̺�����ͭ�Ĵ�Ч�������Ԣ���x����ֵΪ3.0g������������������ʱȢ�죬�ɴ˵ó���ʵ������Ǵ��������������������Һ�ֽ������йأ�
��2������̹ܽ����ܳ������ų��Լ�ƿ�е�ˮ������ѡc���ӱ���ʵ�����ݿ�֪˫��ˮŨ��Խ�ֽ��Խ����
��3�������������Գ��ڵ�������ʣ�����ʵ��������ᣬ����������������ɵ��������������ʿ���ͨ���Ƚ���ͬʱ���ڷ�Ӧǰ����ƽ�Ķ���֮��Ҳ�ܴﵽʵ��Ŀ����