��Ŀ����
ijѧ��Ϊ�˲ⶨʵ������һƿ�治�ƶ����ֱ��ʵ�����������̼���Ƶ��������������������ͼ��ʾ��װ�ã�ͼ������̨����ȥ����ʵ������27�桢110kPa�½��еģ�ʵ�鲽�����£�
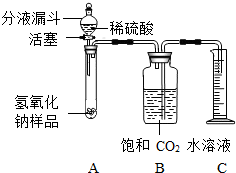
����1����ͼ���Ӻ�װ�ã�
����2������ƽȷ��ȡ����������Ʒ2g����A���Թ��ڣ���B�м���ƿ�ڵ��뱥�Ͷ���̼ˮ��Һ��ƿ������
����3�����Һ©���е���ϡ���ᣬ��������ϡ��������Թ������������رջ�������Ӧ������ͨ��������Ͳ���ռ������Ͷ�����̼ˮ��Һ�����������������220mL��
��1���ж��������Ʒ������ʵ�ʵ��������______���������Ʊ��ʵ�ԭ����______�����ʵ�������������Ʊ���______���森
��2����ʵ�鲽��1�벽��2֮�䣬ȱ��һʵ�鲽�裬��ʵ�鲽����______��
��3��B�м���ƿʢװ���Ͷ�����̼��ˮ��Һ������ˮ���棬��������______��
��4���ж�ʵ�鲽��3�е����ϡ�����ѹ����ı�־��______��
��5������������Ʒ��̼������������Ϊ______����ʱ������̼���ܶ� Ϊ1.8g/L�� ��д��������̣�
��6��������װ�ò��ܲⶨ�Ѳ��ֱ��ʵ�����������Ʒ���������Ƶ�������������������______��

����1����ͼ���Ӻ�װ�ã�
����2������ƽȷ��ȡ����������Ʒ2g����A���Թ��ڣ���B�м���ƿ�ڵ��뱥�Ͷ���̼ˮ��Һ��ƿ������
����3�����Һ©���е���ϡ���ᣬ��������ϡ��������Թ������������رջ�������Ӧ������ͨ��������Ͳ���ռ������Ͷ�����̼ˮ��Һ�����������������220mL��
��1���ж��������Ʒ������ʵ�ʵ��������______���������Ʊ��ʵ�ԭ����______�����ʵ�������������Ʊ���______���森
��2����ʵ�鲽��1�벽��2֮�䣬ȱ��һʵ�鲽�裬��ʵ�鲽����______��
��3��B�м���ƿʢװ���Ͷ�����̼��ˮ��Һ������ˮ���棬��������______��
��4���ж�ʵ�鲽��3�е����ϡ�����ѹ����ı�־��______��
��5������������Ʒ��̼������������Ϊ______����ʱ������̼���ܶ� Ϊ1.8g/L�� ��д��������̣�
��6��������װ�ò��ܲⶨ�Ѳ��ֱ��ʵ�����������Ʒ���������Ƶ�������������������______��
��1�����������ڿ����������տ����е�ˮ�ֶ����⣬Ȼ���������еĶ�����̼��Ӧ�����ʣ������������������ʣ��������̼���ƣ���ô������ϡ����ʱ�����������̼���壬����A�л�ð�����ݣ��ʱ����Ϊ��A�������ݲ�����B���ռ�������/C���ռ���Һ�壩 ������е�CO2��Ӧ �ܷ�
��2����ʵ���������������룬����ʵ��ǰ������װ�õ������ԣ��ʱ����Ϊ�����װ��������
��3����Ӧ������Ҫ���ɶ�����̼����������̼���ˮ��Ӧ��������ˮ�����������ˮ�ᵼ��ʵ������ƫ��ʱ����Ϊ��������̼������ˮ
��4�����������Ʊ��ʣ�������ϡ����ʱ����������壬�����ٲ�������ʱ��˵����Ӧ��ϣ��ʱ����Ϊ������ϡ���ᣬA�в��������ݲ���
��5��47.7%
CO2��������m��CO2��=220mL��1000��1.8g/L=0.396g
����Ʒ��̼��������Ϊx
Na2CO3 +2HCl�T2NaCl+H2O+CO2��
106 44
X 0.396g
=
����ã�X=0.954g
��Ʒ��̼���Ƶ���������Ϊ��
��100%=47.7%
��6�����������ڿ����������տ����е�ˮ�ֶ����⣬Ȼ��Ż�������еĶ�����̼��Ӧ�����ʣ�����û�в����ˮ�������������е����������Բ���������Ƶ������������ʱ����Ϊ������������Ʒ�г���̼���ƣ�����ˮ
��2����ʵ���������������룬����ʵ��ǰ������װ�õ������ԣ��ʱ����Ϊ�����װ��������
��3����Ӧ������Ҫ���ɶ�����̼����������̼���ˮ��Ӧ��������ˮ�����������ˮ�ᵼ��ʵ������ƫ��ʱ����Ϊ��������̼������ˮ
��4�����������Ʊ��ʣ�������ϡ����ʱ����������壬�����ٲ�������ʱ��˵����Ӧ��ϣ��ʱ����Ϊ������ϡ���ᣬA�в��������ݲ���
��5��47.7%
CO2��������m��CO2��=220mL��1000��1.8g/L=0.396g
����Ʒ��̼��������Ϊx
Na2CO3 +2HCl�T2NaCl+H2O+CO2��
106 44
X 0.396g
| 106 |
| x |
| 44 |
| 0.396g |
��Ʒ��̼���Ƶ���������Ϊ��
| 0.954g |
| 2g |
��6�����������ڿ����������տ����е�ˮ�ֶ����⣬Ȼ��Ż�������еĶ�����̼��Ӧ�����ʣ�����û�в����ˮ�������������е����������Բ���������Ƶ������������ʱ����Ϊ������������Ʒ�г���̼���ƣ�����ˮ

��ϰ��ϵ�д�
�����Ŀ
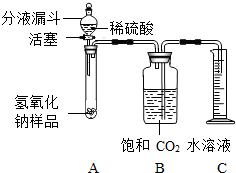
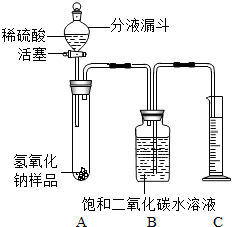 ijѧ��Ϊ�˲ⶨʵ������һƿ�治�ƶ����ֱ��ʵ�����������̼���Ƶ������������������ͼ��ʾ��װ�ã�ͼ������̨����ȥ����ʵ����27�棬101kPa�½��У�ʵ�鲽�����£�
ijѧ��Ϊ�˲ⶨʵ������һƿ�治�ƶ����ֱ��ʵ�����������̼���Ƶ������������������ͼ��ʾ��װ�ã�ͼ������̨����ȥ����ʵ����27�棬101kPa�½��У�ʵ�鲽�����£� ijѧ��Ϊ�˲ⶨʵ������һƿ�治�ƶ����ֱ��ʵ�����������̼���Ƶ������������������ͼ��ʾ��װ�ã�ͼ������̨�Ѿ���ȥ����ʵ����27�棬101kPa�½��У�ʵ�鲽�����£�
ijѧ��Ϊ�˲ⶨʵ������һƿ�治�ƶ����ֱ��ʵ�����������̼���Ƶ������������������ͼ��ʾ��װ�ã�ͼ������̨�Ѿ���ȥ����ʵ����27�棬101kPa�½��У�ʵ�鲽�����£�