��Ŀ����
����Cu��CuO�Ĺ�������20g��Ϊ�˷����������Cu��CuO�ĺ�����С
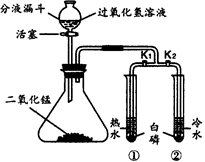
��ͬѧ����ϡ�����п����ȡ�������������ͼ��ʾ��װ�ã������йص�ʵ��̽����

�õ��������ݣ����ٶ�ÿ������ȫ��Ӧ�����գ�
����ʵ����Ƽ��й����ݽ��з�������㣺
��1������ˮ�������� g��
��2���������CuO������������Ϊ���٣���д��������̣�
��3����ʵ�黹�ɲⶨ���ˮ�ĸ�Ԫ��֮���������ϵ�����ñ���ʵ��������ʽ��ʾ��ˮ���⡢��Ԫ�ص�������Ϊ_______________________________________�����û������������е�����ͭû����ȫ��Ӧ�������ˮ���⡢��Ԫ�ص������Ȼ�__________��ѡ�ƫ����ƫС�� ���䡱����
��ͬѧ����ϡ�����п����ȡ�������������ͼ��ʾ��װ�ã������йص�ʵ��̽����

�õ��������ݣ����ٶ�ÿ������ȫ��Ӧ�����գ�
| װ�� | B | C |
| ��Ӧǰ���� | 85.3g | 284.2g |
| ��Ӧ������ | 82.1g | 287.8g |
����ʵ����Ƽ��й����ݽ��з�������㣺
��1������ˮ�������� g��
��2���������CuO������������Ϊ���٣���д��������̣�
��3����ʵ�黹�ɲⶨ���ˮ�ĸ�Ԫ��֮���������ϵ�����ñ���ʵ��������ʽ��ʾ��ˮ���⡢��Ԫ�ص�������Ϊ_______________________________________�����û������������е�����ͭû����ȫ��Ӧ�������ˮ���⡢��Ԫ�ص������Ȼ�__________��ѡ�ƫ����ƫС�� ���䡱����
��1����1�֣�3.6
��2����4�֣������1�֣�������3�֣�
�⣺��������CuO������ΪX
H2+CuO Cu+H2O
Cu+H2O
80 18
X 3.6g
80��18= X��3.6g
��� X="16" g
������������˵���������Ϊ�� ��100%=80%
��100%=80%
�𣺻����������ͭ������������80%
��3����2�֣�H��O=[��287.8��284.2������85.3-82.1��]����85.3-82.1��
��2����4�֣������1�֣�������3�֣�
�⣺��������CuO������ΪX
H2+CuO
 Cu+H2O
Cu+H2O80 18
X 3.6g
80��18= X��3.6g
��� X="16" g
������������˵���������Ϊ��
 ��100%=80%
��100%=80%�𣺻����������ͭ������������80%
��3����2�֣�H��O=[��287.8��284.2������85.3-82.1��]����85.3-82.1��
�����������1����ͼʾװ���У�A�������dz�ȥ�����е�ˮ������B�������Ƿ�Ӧװ�ã�C���������������ɵ�ˮ����������ˮ������ʵ���Ͼ���Cװ�õ������仯ֵ����287.8g��284.2g=3.6g
��2����֪��ˮ������ δ֪������ͭ�ڻ�����е����������� ����˼·���ɸ���ˮ������ͭ�ڷ�Ӧ�е�������ϵ�������ͭ����������������������������������̼���
��3��װ��B�ڵ������ڷ�ӦǰΪ����ͭ����Ӧ��Ϊͭ�����Է�Ӧǰ���������ֵ��Ϊ��Ԫ�ص�����������85.3-82.1��g��ˮ����Ԫ�ص�����Ϊ��[��287.8��284.2������85.3-82.1��]g��

��ϰ��ϵ�д�
 �������ͬ������ϵ�д�
�������ͬ������ϵ�д�
�����Ŀ