��Ŀ����
��ѧ��һ����ʵ��Ϊ��������Ȼѧ�ƣ����л�ѧ������������ʵ�飬���磺

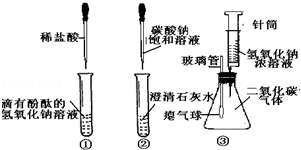
��1��A����̽���������ʵ�ʵ�飬ͨ��ʵ���֪��ʽ̼��ͭ���еĻ�ѧ�������� ��������ȷ��ʵ����������ܻ��ʵ�����Ӱ���������غ���������̽�������Ŀ�ȼ��ʱ��Ϊ�˱�֤��ȫ��������е�һ��������� ��
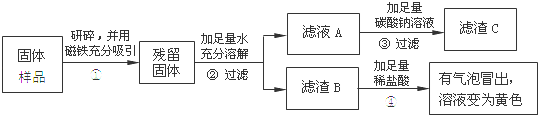
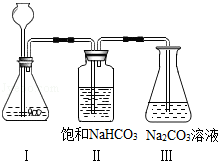
��2��B����̽����ѧ��Ӧ����������һ��ʵ�飬�ܶѧ��Ӧ�ķ�������Ҫ����һ����������ͼ����ʾʵ�������������ȼ����Ҫ��������������Ƶ�һ������ �������⣬���ǻ�����̽�������ᡢ�������Һ�з������ֽ���������� ��

��1��A����̽���������ʵ�ʵ�飬ͨ��ʵ���֪��ʽ̼��ͭ���еĻ�ѧ�������� ��������ȷ��ʵ����������ܻ��ʵ�����Ӱ���������غ���������̽�������Ŀ�ȼ��ʱ��Ϊ�˱�֤��ȫ��������е�һ��������� ��
��2��B����̽����ѧ��Ӧ����������һ��ʵ�飬�ܶѧ��Ӧ�ķ�������Ҫ����һ����������ͼ����ʾʵ�������������ȼ����Ҫ��������������Ƶ�һ������ �������⣬���ǻ�����̽�������ᡢ�������Һ�з������ֽ���������� ��
��1���������ֽ⣻���������Ĵ��ȣ���2���������Ӵ����г���������������ų�����ˮ����
�����������1����ʽ̼��ͭ���ȷֽ������ͭ��ˮ�Ͷ�����̼��˵���˼�ʽ̼��ͭ�����ֽ�Ļ�ѧ���ʣ���ȼ��ȼ����������������������Ļ������������ը���������п�ȼ�ԣ����Ե�ȼǰҪ���������Ĵ��ȣ���2�������������������������ˮ��ͬ�Ӵ�������ȼ�յ������ǣ����п�ȼ�ԡ��������Ӵ����¶ȴﵽ��ȼ����Ż�㣬���������������ȼ����Ҫ��������������Ƶ�һ���Ƕ��������Ӵ������ֽⷴӦ��Ҫ������������г���������������ų�����ˮ����

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ