��Ŀ����
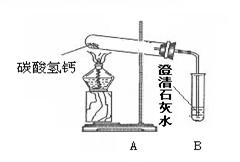
ij�С��ͬѧ�ü��Ȳ��ᾧ���Ƶõ�CO��H2C2O4��2 H2O==CO��+ CO2��+3H2O����������ԭ��ʵ�鲢����β���������������װ�ã�ͼ�ס�ͼ�ң����ش������й����⣺

��1���ڼ��ȷֽ���ᾧ���װ���У�������������ѡ��ʹ�ã�
���Թܢ��ձ��۾ƾ��Ƣ������������̨�������У������ܵ���Ƥ������ƿ
������Ҫ�õ������� ������ţ�
��2��Ϊ�˻�ø��﴿����CO��A��B��װ����Ӧ�ֱ��� ��
��3��Ӳ�ʲ������з�����Ӧ�Ļ�ѧ����ʽ
��4��ѡ��ͼ�״���β����ʵ��ʱӦ�ȵ�ȼ ���a����b�������ľƾ��ơ�ͼ����ͼ����ʾ��β������������Ƚϣ�����Ҫ�ŵ���
��5���С���ͬѧͨ���������ϵ�֪�������£�CO�����Ȼ���PdCl2��Һ��Ӧ����������ѡ���˽�β��ֱ��ͨ���Ȼ�����Һ�н��д������÷�Ӧ�Ļ�ѧ����ʽΪ��CO+ PdCl2+H2O==Pd��+ CO2 + 2R,����R�Ļ�ѧʽΪ ���˷�������������ȳ��˾���ͼ�ұ�ͼ����ʾ�������ŵ��⣬��ʵ��Ƕȿ������е��ŵ���
��д��һ�������𰸼��ɣ�

��1���ڼ��ȷֽ���ᾧ���װ���У�������������ѡ��ʹ�ã�
���Թܢ��ձ��۾ƾ��Ƣ������������̨�������У������ܵ���Ƥ������ƿ
������Ҫ�õ������� ������ţ�
��2��Ϊ�˻�ø��﴿����CO��A��B��װ����Ӧ�ֱ��� ��
��3��Ӳ�ʲ������з�����Ӧ�Ļ�ѧ����ʽ
��4��ѡ��ͼ�״���β����ʵ��ʱӦ�ȵ�ȼ ���a����b�������ľƾ��ơ�ͼ����ͼ����ʾ��β������������Ƚϣ�����Ҫ�ŵ���
��5���С���ͬѧͨ���������ϵ�֪�������£�CO�����Ȼ���PdCl2��Һ��Ӧ����������ѡ���˽�β��ֱ��ͨ���Ȼ�����Һ�н��д������÷�Ӧ�Ļ�ѧ����ʽΪ��CO+ PdCl2+H2O==Pd��+ CO2 + 2R,����R�Ļ�ѧʽΪ ���˷�������������ȳ��˾���ͼ�ұ�ͼ����ʾ�������ŵ��⣬��ʵ��Ƕȿ������е��ŵ���
��д��һ�������𰸼��ɣ�
��1���٢ۢݢ�
��2��NaOH��Һ��ŨH2SO4 ��3��3CO+Fe2O3 2Fe+3CO2
2Fe+3CO2
��4��b������Լ��Դ���𰸺������ɣ� ��5��HCl����ȫ�����������У��������ɣ�
��2��NaOH��Һ��ŨH2SO4 ��3��3CO+Fe2O3
 2Fe+3CO2
2Fe+3CO2��4��b������Լ��Դ���𰸺������ɣ� ��5��HCl����ȫ�����������У��������ɣ�
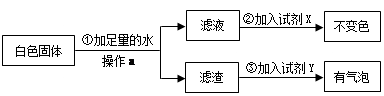
��1��Ҫ���Ȳ��ᾧ����Ҫ��֧�ż��ȵ�װ�ã���Ҫ��ʢ�Ų����װ�ã���Ҫ����װ�ã���Ҫ����װ�ã��ʴ�Ϊ���٢ۢݢޣ�
��2�����Ⱥ������ˮ�Ͷ�����̼��һ����̼����Ҫ��������̼��ˮ��ȥ����ȥˮ��Ũ���ᣬ��ȥ������̼���������ƣ�ˮҪ���ں����ȥ���ʴ�Ϊ��NaOH��Һ��ŨH2SO4��
��3��һ����̼���������ڸ��µ��������������Ͷ�����̼���ʷ���ʽΪ��3CO+Fe2O3 2Fe+3CO2��
2Fe+3CO2��
��4��һ����̼���п�ȼ�ԣ�����ȵ�ȼa���ľƾ��ƣ�һ����̼�������Ļ����������ʱ�ᷢ����ը���ʴ�Ϊ��b��
ͼ���е�һ����̼�ֵõ������ã�������ȼ���ṩ�������ʴ�Ϊ����Լ��Դ��
��5�����������غ㶨�ɿ�֪����Ӧǰ���Ԫ�������ԭ�Ӹ������䣬�ʴ�Ϊ��HCl��
�ò�������Ҫͼ�ס�ͼ���е�̫��װ�ã�Ҳ����Ҫ��ȼ�������˱�ը��Σ�գ��ʴ�Ϊ����ȫ�����������У�
��2�����Ⱥ������ˮ�Ͷ�����̼��һ����̼����Ҫ��������̼��ˮ��ȥ����ȥˮ��Ũ���ᣬ��ȥ������̼���������ƣ�ˮҪ���ں����ȥ���ʴ�Ϊ��NaOH��Һ��ŨH2SO4��
��3��һ����̼���������ڸ��µ��������������Ͷ�����̼���ʷ���ʽΪ��3CO+Fe2O3
 2Fe+3CO2��
2Fe+3CO2����4��һ����̼���п�ȼ�ԣ�����ȵ�ȼa���ľƾ��ƣ�һ����̼�������Ļ����������ʱ�ᷢ����ը���ʴ�Ϊ��b��
ͼ���е�һ����̼�ֵõ������ã�������ȼ���ṩ�������ʴ�Ϊ����Լ��Դ��
��5�����������غ㶨�ɿ�֪����Ӧǰ���Ԫ�������ԭ�Ӹ������䣬�ʴ�Ϊ��HCl��
�ò�������Ҫͼ�ס�ͼ���е�̫��װ�ã�Ҳ����Ҫ��ȼ�������˱�ը��Σ�գ��ʴ�Ϊ����ȫ�����������У�

��ϰ��ϵ�д�
 ��������ϵ�д�
��������ϵ�д�
�����Ŀ