��Ŀ����
����Ŀ��ij�о���ѧϰС������˲ⶨ���Na2CO3����Ʒ��ֻ��NaCl���ʣ���Na2CO3����������ʵ�鷽������ش��������⣺
С���ϣ���ʯ�ҵ���Ҫ�ɷ����������ƺ������ƵĻ����
��1�����巨��
��ԭ��˼·������Na2CO3��ϡ���ᷴӦ����CO2��ͨ��������װ��ʵ��ǰ��������ó�CO2�������Ӷ�����̼���Ƶ�������������������ԭװ���ڿ�����Ӱ�죩
��ʵ�������С��ͬѧ��������˼·���������ͼ1��ʵ��װ�á�
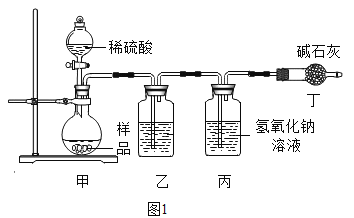
��ʵ��װ������ʢ�ŵ��Լ�Ϊ______________��
��������___________________________________��
װ�ö��м�ʯ�ҵ�������________��������ĸ��
A������Na2CO3��ϡ���ᷴӦ����CO2
B����ֹ�������ж�����̼�������
C�����������̼
��ʵ��������μ�ϡ������ٶȹ��죬�ᵼ�²����Ʒ��̼���Ƶ�
��������______����д��ƫ������ƫ������������������
��С��ͬѧ��ΪС�Ƶķ����������⣬��ʹ�����淶��Ҳ����ɽ��ƫ�ͣ���
��Ϊ����������_______________________________��Ӧ�ý�װ�ü���˫����
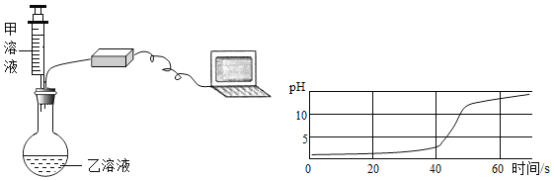
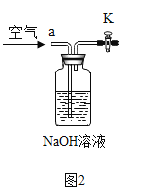
������������������������ͼ2װ�ã�ʵ�������ͨ��һ��ʱ�������
Ŀ����___________________������NaOH��Һ��������_________________��
��2����������
��ʵ��ԭ��������̼�������������ӽ�����ɳ�����ͨ���������������ó�̼���Ƶ�����������
��ʵ�鲽�裩����������ƽ��ȡ��Ʒ11��0g�����ձ��У���ˮʹ����ȫ�ܽ⣻
�����������CaCl2��Һ��ַ�Ӧ��֤��CaCl2��������Na2CO3����ȫ��Ӧ���ķ�����: ���ã����ϲ���Һ�еμ�___________��Һ�����û�а�ɫ�������ɣ�֤��CaCl2������
�����ˡ�����X�������������������Ϊ10��0g������X��������_______�����ȱ�ٴ˲��������̼������������__________����д��ƫ������ƫ������������������
��ͨ�������ṩ�����ݼ������Ʒ��̼���Ƶ���������Ϊ________����д��������̣����������0��1%����3�֣�
���𰸡�Ũ���ᣨ��ŨH2SO4�� ����ˮ�����������CO2�� B ƫ�� ��CO2��������ƿ�� ��������CO2ȫ�����뵽��װ���У�ʹ����ȫ������������Һ���� ���տ����е�CO2 CaCl2 ϴ�� ƫ�� 96��4%
��������
��1��
����Ϊ��ͨ��������װ��ʵ��ǰ��������ó�CO2���������Զ�����̼����������dz�ȷ�����Եñ�֤���뵽װ�ñ����Ǵ����Ķ�����̼��������ʵ��װ������ʢ�ŵ��Լ�Ϊ��Ũ���ᣨ��ŨH2SO4���������ǣ�����ˮ�����������CO2����װ�ö��м�ʯ�ҵ������ǣ���ֹ�������ж�����̼������У���ѡB��
��ʵ��������μ�ϡ������ٶȹ��죬�����Ķ�����̼��������Ӧ�����ų���װ�ñ��⣬ʹ�ö�����̼������ƫС����Ȼ�ᵼ�²����Ʒ��̼���Ƶ���������ƫ�ͣ�
��С��ͬѧ��ΪС�Ƶķ����������⣬��ʹ�����淶��Ҳ����ɽ��ƫ�ͣ����������ǣ���CO2��������ƿ�ڣ�Ӧ�ý�װ�ü���˫��������������������������ͼ2װ�ã�ʵ�������ͨ��һ��ʱ�������Ŀ���ǣ���������CO2ȫ�����뵽��װ���У�ʹ����ȫ������������Һ���գ�����NaOH��Һ�������ǣ����տ����е�CO2����ֹʹ������̼������ƫ��
��2��
�����������CaCl2��Һ��ַ�Ӧ��֤��CaCl2��������Na2CO3����ȫ��Ӧ���ķ�����: ���ã����ϲ���Һ�еμ�Na2CO3��Һ�����û�а�ɫ�������ɣ�֤��CaCl2������
�����ˡ�����X�������������������Ϊ10��0g������X��������ϴ�ӣ���ֹ������ճ��NaCl�����ȱ�ٴ˲��������̼��������ƫ����������ƫ�ߣ�
��������Ӧ�ķ���ʽΪ��CaCl2+Na2CO3=CaCO3��+2NaCl������������Ϊ10��0g����̼��Ƶ����������ݷ���ʽ��Na2CO3��̼��Ƶ�������ϵ���������Na2CO3����������һ�������Ʒ��̼���Ƶ���������
�⣺����Ʒ��̼���Ƶ�����Ϊx��
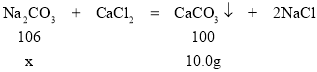
![]()
![]()
x=10��6g
����̼���Ƶ�����������![]() ��100%��96��4%��
��100%��96��4%��

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�