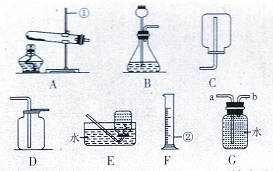
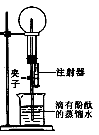
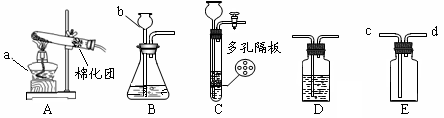
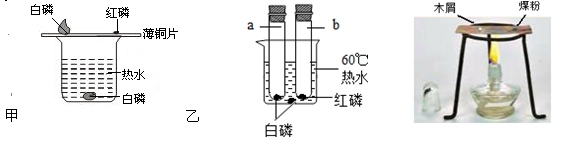
��Ŀ����
ͨ��ѧϰ������ʣ���У��ѧ��ȤС���ͬѧ��ʵ���ҵġ����������Ƿ�����Լ���ֵij̶ȡ�չ����̽���������һͬ���룺
��������⣺���������Ƿ���ʣ�
�Ʋ�������裺ͬѧ�Ǿ���������Ϊ��Ʒ�������ֿ��������û�б��ʣ����ֱ��ʣ���ȫ���ʡ�
(3)ʵ��̽��
(4)С����˼����
��С��ͬѧ��������ʵ�鷽������ʵ�飬ȷ������Ʒ�Ѳ��ֱ��ʣ���д��ʵ���з�Ӧ�Ļ�ѧ����ʽ��
�����������׳���ʯ�ң�ũҵ�ϳ����� ��
��������⣺���������Ƿ���ʣ�
�Ʋ�������裺ͬѧ�Ǿ���������Ϊ��Ʒ�������ֿ��������û�б��ʣ����ֱ��ʣ���ȫ���ʡ�
(3)ʵ��̽��
| ʵ�鲽�輰���� | ʵ������ | ʵ����� |
| ȡ�����Թ��У�������������ˮ������ ��ȡ�ϲ���Һ��������ɫ��̪��Һ �ڵ�ȥ�ϲ���Һ�������Թ���ע��ϡ���� | ����ɫ��̪��Һ��� �� | ���ֱ��� |
| ����ɫ��̪��Һ����� �� | | |
| �� ��û�����ݲ��� | |
(4)С����˼����
��С��ͬѧ��������ʵ�鷽������ʵ�飬ȷ������Ʒ�Ѳ��ֱ��ʣ���д��ʵ���з�Ӧ�Ļ�ѧ����ʽ��
�����������׳���ʯ�ң�ũҵ�ϳ����� ��
(3)ʵ��̽����
(4)С����˼����
Ca(OH)2+CO2 = CaCO3��+H2O CaCO3+2HCl = CaCl2+H2O+CO2��
�ڸ�����������
| ʵ�鲽�輰���� | ʵ������ | ʵ����� |
| | �� �������ݲ��� | |
| �� �������ݲ��� | ȫ������ | |
| ����ɫ��̪��Һ��� �� | û�б��� |
Ca(OH)2+CO2 = CaCO3��+H2O CaCO3+2HCl = CaCl2+H2O+CO2��
�ڸ�����������
�����������3�����ݽ��۷��������ֱ��ʣ�������Ʒ�к����������ƺ�̼��ƣ��ʢڵ�ȥ�ϲ���Һ�������Թ���ע��ϡ�����϶������ݲ�����̼��ƺ�ϡ���ᷴӦ�������Ȼ��ơ�ˮ�Ͷ�����̼����
���������������ȡ�ϲ���Һ��������ɫ��̪��Һ����ɫ��̪��Һ����죬˵���������������ƣ���һ��ȫ��̼��ƣ�˵��ҩƷȫ�����ʣ����Ԣڵ�ȥ�ϲ���Һ�������Թ���ע��ϡ���ᣬ�϶������ݲ�����
��������������ڵ�ȥ�ϲ���Һ�������Թ���ע��ϡ���ᣬû�����ݲ�����˵��û��̼��ƣ���ֻ���������ƣ�˵��ҩƷû�б��ʣ����Ԣ�ȡ�ϲ���Һ��������ɫ��̪��Һ����ɫ��̪��Һ��졣
��4���ٸ������⣬���ֱ��ʣ�������Ʒ�к����������ƺ�̼����������ʣ�����̼��������������Ʊ������ɣ���������̼���������Ʒ�Ӧ����̼��ƺ�ˮ���ʷ�Ӧ�Ļ�ѧ����ʽΪCa(OH)2+CO2 = CaCO3��+H2O���������⣬֤���������Ʊ��ʵ����������Թ���ע��ϡ����������ݲ�������̼��ƺ�ϡ���ᷴӦ�������Ȼ��ơ�ˮ�Ͷ�����̼���ʷ�Ӧ�Ļ�ѧ����ʽΪCaCO3+2HCl = CaCl2+H2O+CO2����
�����������׳���ʯ�ң�ũҵ�ϳ�����ũҵ��������������������
������������֤��ʵ��̽����Ҫ������ʵ����ʻ�仯���ɣ����ݸ�����ʵ����Ʒ���������ʵ�顢������̽������ͨ���۲졢��¼�ͷ�����ʵ����������֤�����ʵ����ʻ�仯���ɵȡ�

��ϰ��ϵ�д�
�����Ŀ