��Ŀ����
����Ŀ����������ʵ��װ��ͼ���ش��й����⡣
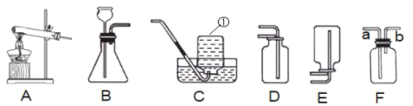
��1��д��ָ�����������ƣ���______________��
��2��ʵ�����ø��������ȡ���ռ��ϴ�������������ѡ�õķ���װ����______________������ĸ��ţ���װ��ҩƷ�������ӵ���Ʒ��____________����������_____________________����Ӧ�ķ��ű���ʽΪ___________________________________����ѡ��Cװ���ռ����������䴿������ƫ�ͣ�ԭ�������_________������ţ�
�ٸ�������л����˶������� ���ռ�ǰ������ƿ��δװ��ˮ
���ռ�����ƿ����������ˮ �ܵ����ܿڸ�������ð���������ռ�
��3�����⣨H2S��������һ����ɫ�г�������ζ�����壬�ܶȱȿ�����������ˮ�� ʵ�����п����ù�����������FeS����ϡ�����ڳ����·�Ӧ��ȡ��ʵ������ȡ��������ѡ�õķ���װ��Ϊ__________������ĸ��ţ��������Fͼ��ʾװ���ռ��������壬����Ӧ��___________���a����b�����˹ܿ�ͨ�롣
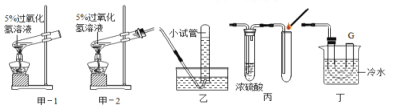
��4��������ʾ�������Ƶĺ�H2O2 5%����H2O2������������Һ��������100%������Һ�����ȵ�80��ʱ�����н϶���������������ͬ������H2O25%����Һ��������������¾ͻ�����������������Ӧ�ٶȿ죬����ʱ��̡�С����ͼ��-1װ�ý���ʵ�飬���Թ����д������ݲ����д�����������ʱ���ô����ǵ�ľ�����ڵ��ܿڣ�ľ����δ��ȼ��Ϊ�ˣ������Һͼ�-2����������ͼ��װ���ռ����壬���ô����ǵ�ľ�����飬ľ����ȼ����ôͼ��ʵ���д�����ľ��δ��ȼ��ԭ����________________________����ʦ���������һ���Ľ��ı�װ�ã������ͼ�-2�������ô����ǵ�ľ�����飬ľ��Ҳ��ȼ����ͨ������˵��Ũ���������__________________�������ͼ�-2�������ô����ǵ�ľ����G�����飬ľ��____________�����ܻ��ܣ���ȼ��
���𰸡�����ƿ A �� ��ֹ����ʱ������ط�ĩ���뵼���� KMnO4![]() K2MnO4+MnO2+O2 �ڢ� B a �����л��н϶��ˮ������������Ũ�ȵ͡�ˮ�����������ߵȣ��������ɣ� ����ˮ���� ��
K2MnO4+MnO2+O2 �ڢ� B a �����л��н϶��ˮ������������Ũ�ȵ͡�ˮ�����������ߵȣ��������ɣ� ����ˮ���� ��
��������
��1������1�������Ǽ���ƿ��
��2��ʵ�����ø��������ȡ���ռ��ϴ���������ʱ����Ӧ���״̬�ǹ�̬����Ӧ�������Ǽ��ȣ��ʷ���װ��ѡ�̹̼�����װ��A��������طֽ�����ʹ��죬Ϊ�˷�ֹ����ʱ������ط�ĩ���뵼������װ��ҩƷ�������ӵ���Ʒ�������÷�Ӧ�ķ��ű���ʽ�ǣ�KMnO4![]() K2MnO4+MnO2+O2���ٸ�������л����˶������̲��ᵼ���Ƶõ������������ʴ��������ռ�ǰ������ƿ��δװ��ˮ���ͻ��в��ֿ�����ʹ�����ռ������岻��������ȷ�����ռ�����ƿ����������ˮ���ᵼ���ռ������岻�����ʴ��ܵ����ܿڸ�������ð���������ռ��������ռ��������л���п���������ȷ����ѡ�ڢܡ�
K2MnO4+MnO2+O2���ٸ�������л����˶������̲��ᵼ���Ƶõ������������ʴ��������ռ�ǰ������ƿ��δװ��ˮ���ͻ��в��ֿ�����ʹ�����ռ������岻��������ȷ�����ռ�����ƿ����������ˮ���ᵼ���ռ������岻�����ʴ��ܵ����ܿڸ�������ð���������ռ��������ռ��������л���п���������ȷ����ѡ�ڢܡ�
��3��ʵ������ȡ��������ķ�Ӧ���״̬�ǹ����Һ�壬��Ӧ�������Dz����ȣ���ѡ��Һ��������װ��B��Fװ���Ƕ��װ�ã��������ռ����⣬����������ܶȱȿ������ܶȴ����������ɳ���aͨ�롣
��4�����ȹ���������80���϶Ȳ��н϶���������ɣ���ʱ�ռ��������л��н϶��ˮ�������ʴ����ǵ�ľ�����ܸ�ȼ��ͼ����Ũ���Ὣ�����ˮ�������գ��������Ĵ������ӣ��ʸ�ȼ��������徭��U�ܺ�ˮ����Ԥ��Һ�������ռ��������ϴ����ʿ��Ը�ȼ��

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�����Ŀ��ͬѧ�ǿ���ͨ�����з�ʽ��ʶ������
����ɽǶȣ�
�ڵ�����������������������̼���������������С�����ѧ������д��
���ǿ�������ɳɷֻ����������������__�������ڴ�����������������_____������ʱ�̶��벻������������Ϊ��_____�� ����������ЧӦ��������_____, д��ʵ���Ҽ��������Ļ�ѧ����ʽ_________
��Ϊ�ⶨ������������������������ͼʵ�顣
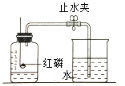
I����ʵ���к�����Ҫ������ԭ����__________��
II������ȼ�յ�������_________����Ӧ�Ļ�ѧ����ʽ_____________��
III����ȴ�����º��ֹˮ�й۲쵽��������_______��
ijͬѧ�ø�װ�����ⶨ�����������������ʵ��ʱ�������������趼��ȷ����ֹˮ��û�н��������ⶨ���������������� ____���� ƫ���ƫС��Ӱ�죩
���۽Ƕ� ��![]()
���á���ѧ���š���ͼʾ����ա�
ͼ ʾ |
| ______ |
|
��ѧ���� | ___ | N2 | _____ |
��ͬ��ͬѹ�£����������ȵ��ڷ��Ӹ����ȡ������Կ����������ɷ֣���ͼ�ɱ�ʾ������ģ�͵���________����ѡ���
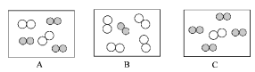
���仯�Ƕȣ�
��һ��������ѹ�£������в�����ֵķе����£�
��� | ���� | ���� | ������̼ |
�е㣨�棩 | -195.8 | -183.0 | -78.4 |
ȼ�ŵ�ľ������ʢ��Һ̬������-200�棩�ĸ�ƿ�ڣ��۲쵽��������____��
�����������������_____��
A��ʳƷ��װ�ڳ�N2�Է�������ΪN2�Ļ�ѧ���ʱȽ��ȶ���
B����ҵ�Ϸ���Һ̬������ȡ�����ǻ�ѧ�仯
C���ӱ������ó���ˮ��ƿ�������Һ�飬˵����������ˮ����
D�����ó���ʯ��ˮ���Լ�ƿ�ڱ���һ���Ĥ��֤���������ж�����̼
����Ŀ��ѧϰ��������֪ʶ����ȤС��ͬѧ�������������е�Ӧ�ý�����̽����
���������ϣ���������������ͼ���Ĺ���ԭ�����£���ѹʱ��װ���������ڲ��ķ���ɸ���������еĵ�������ȡ��Ũ����������ѹʱ������ɸ���������ĵ����ŷţ����������У�����ɸ�������ġ���ȤС��ͬѧ�ü�����������ȡ���ռ���һ�����壬����ʵ���Ҷ�������о���
��һ��ʵ��I��С��ͬѧ��ͨ������ʵ����֤���ռ��������Ƿ�Ϊ������������
��ʵ���¼��ȡһֻ 250mL �ļ���ƿ������ˮ���ռ�һƿ�������壬Ȼ���һ��ȼ�ŵ�ľ�����뼯��ƿ�У�ľ��ȼ�յø�����
��ʵ����ۣ��ռ��������Ǵ�����������
��ʵ�鷴˼����ʦָ������ʵ�鲢����֤��С��ͬѧ������������һ���Ǵ�������������ʵ �鲻��ȷ�����ռ�����Ϊ����������ԭ����_____��
������ʵ�� II���ⶨС��ͬѧ���������������ĺ�����
ͬѧ������ʦ��ָ�����������ͼ��ʾװ�ã�����ʵ�飬���ظ���Ρ��벹����ʵ���¼��
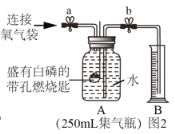
��ʵ�鲽�裩
�ټ��_____��
�ڼ���ҩƷ����װ�����ӹ̶���
�۴�ֹˮ�� a �� b���� A �л�������һ�������壬¶�����ر�ֹˮ��a ��b��A ��ˮ���뵽 B �У����B ��ˮ�����ΪV1mL��
�������۹���ȼ���ף�����ȼ�գ������������̡�
�ݴ�����Ϩ����ȴ������������_____������B ��ˮ�������뵽A ���� �����Ͳ�е������ΪV2mL��
����С��ͬѧ�������������������������
��1�������й����ʵ���˵���У���ȷ������______��
A.������У�������Ͳ������ʹ��õ� V1 ��ֵƫ�ͣ�
B.������У�����ȼ��ʱҪ���ֹر�ֹˮ�� a �� b��
C.������У�δ��ȴ�����»�ʹ��õ�С��ͬѧ���������������ĺ���ƫ�ߣ�
D.���������������������������![]()
��2�������ݼ�¼��
���ⶨ V1=210mL��V2 �������£�
ʵ����� | 1 | 2 | 3 | 4 | 5 |
B ��ʣ��ˮ���/mL | 100 | 52 | 49 | 51 | 48 |
�����ݴ�������֪����������������� 1 ������ƫ��ϴ���������������������ݴ���ʱӦɾȥ��С��ͬѧ�����������������������Ϊ_____%������С�����һλ��Ч���֣�
��3����ʵ�鷴˼��
�ټ����������������Ĺ��̷����ı仯����_____�����������仯��������ѧ�仯������
���ø��������ȡ�����Ļ�ѧ����ʽ_____��������ȡ�ɱ��ϸ������ʺϴ���������һ������ʵ������ȡ��
�۵��µ� 1 �����ݲ����ϴ����Ŀ���ԭ����_____��


