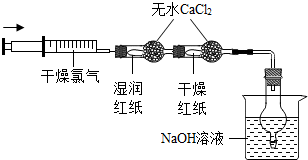
��Ŀ����
���ұ�����ʵ��С��ֱ���С�Na2CO3��NaOH�������Na2CO3�����ⶨ����ʵ�飺
��1�������ʵ�鷽���ǣ���50�˵Ļ�����ܽ���ˮ�������Һ���μ�10%ϡ���ᣬ�۲쵽���� ����������������Һ���������ⶨ̼���Ƶĺ���������Ϊ��������Ƿ���ȷ�������� ��
��2������ͬѧ�ķ����ǣ����ݳ�������������ó�̼���Ƶĺ�������ͼ1
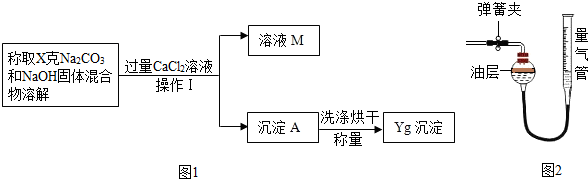
�Ҿ���ȷ���㣬�ó�̼���Ƶĺ���ƫ��ԭ������� ����ҺM�е����ʣ��û�ѧʽ��ʾ���� ��
��3�������ʵ�鷽���ǣ���50����Ʒ��������ϡ���ᷴӦ������ͼ2װ�òⶨ������CO2����������ͨ������ó���Ʒ��Na2CO3�������������װ�����Ͳ�������� �����ռ���0.1Ħ��CO2���壬��ԭ�������Na2CO3�������ٷ����Ƕ��٣�����ȷ��0.1%���������ݻ�ѧ����ʽ��д��������̣�
��1�������ʵ�鷽���ǣ���50�˵Ļ�����ܽ���ˮ�������Һ���μ�10%ϡ���ᣬ�۲쵽����
��2������ͬѧ�ķ����ǣ����ݳ�������������ó�̼���Ƶĺ�������ͼ1

�Ҿ���ȷ���㣬�ó�̼���Ƶĺ���ƫ��ԭ�������
��3�������ʵ�鷽���ǣ���50����Ʒ��������ϡ���ᷴӦ������ͼ2װ�òⶨ������CO2����������ͨ������ó���Ʒ��Na2CO3�������������װ�����Ͳ��������
���㣺ʵ��̽�����ʵ���ɳɷ��Լ�����,��Ļ�ѧ����,�εĻ�ѧ����,���ݻ�ѧ��Ӧ����ʽ�ļ���
ר�⣺��ѧ̽��
��������1����������Ҫ�Ⱥ�����������Һ��Ӧ�����Ȼ��ƺ�ˮ��������������Һ��Ӧ��������̼������Һ��Ӧ�����Ȼ��ơ�ˮ�Ͷ�����̼���н��
��2������̼���ƺ��Ȼ�����Һ��Ӧ����̼��Ƴ��������Ȼ��ƣ�����̼��Ƶ�����������ô�ó�̼���Ƶĺ���ƫ����н��
��3�����ݶ�����̼������ˮ���ܺ�ˮ��Ӧ�Լ�������̼���������̼���Ƶ��������н��
��2������̼���ƺ��Ȼ�����Һ��Ӧ����̼��Ƴ��������Ȼ��ƣ�����̼��Ƶ�����������ô�ó�̼���Ƶĺ���ƫ����н��
��3�����ݶ�����̼������ˮ���ܺ�ˮ��Ӧ�Լ�������̼���������̼���Ƶ��������н��
����⣺��1������Ҫ�Ⱥ�����������Һ��Ӧ�����Ȼ��ƺ�ˮ��������������Һ��Ӧ��������̼������Һ��Ӧ�����Ȼ��ơ�ˮ�Ͷ�����̼�����Խ�50�˵Ļ�����ܽ���ˮ�������Һ���μ�10%ϡ���ᣬ�۲쵽����һ����������ݣ���Ϊ̼���ƺ��������ƶ����������ᣬ���Բ��ܸ�������������Һ���������ⶨ̼���Ƶĺ����������һ����������ݣ�����ȷ����Ϊ̼���ƺ��������ƶ����������
��2��̼���ƺ��Ȼ�����Һ��Ӧ����̼��Ƴ��������Ȼ��ƣ�����̼��Ƶ�����������ô�ó�̼���Ƶĺ���ƫ��ԭ�������ϴ�Ӳ���ֻ�û����ȫ�����Ϊ�ǹ������Ȼ�����Һ��������ҺM�е��������Ȼ��ơ��������ƺ��Ȼ��ƣ����ϴ�Ӳ���ֻ�û����ȫ���CaCl2��NaOH��NaCl��
��3��������̼������ˮ���ܺ�ˮ��Ӧ������װ�����Ͳ�������Ƿ�ֹ������̼�ܽ���ˮ�л��ˮ��Ӧ�������ֹ������̼�ܽ���ˮ�л��ˮ��Ӧ��
��ԭ�������Na2CO3������Ϊx��������̼������=0.1mol��44g/mol=4.4g
Na2CO3+2HCl=2NaCl+H2O+CO2��
106 44
x 4.4g
=
x=10.6g
ԭ�������Na2CO3�������ٷ���=
��100%=21.2%
��ԭ�������Na2CO3�������ٷ�����21.2%��
��2��̼���ƺ��Ȼ�����Һ��Ӧ����̼��Ƴ��������Ȼ��ƣ�����̼��Ƶ�����������ô�ó�̼���Ƶĺ���ƫ��ԭ�������ϴ�Ӳ���ֻ�û����ȫ�����Ϊ�ǹ������Ȼ�����Һ��������ҺM�е��������Ȼ��ơ��������ƺ��Ȼ��ƣ����ϴ�Ӳ���ֻ�û����ȫ���CaCl2��NaOH��NaCl��
��3��������̼������ˮ���ܺ�ˮ��Ӧ������װ�����Ͳ�������Ƿ�ֹ������̼�ܽ���ˮ�л��ˮ��Ӧ�������ֹ������̼�ܽ���ˮ�л��ˮ��Ӧ��
��ԭ�������Na2CO3������Ϊx��������̼������=0.1mol��44g/mol=4.4g
Na2CO3+2HCl=2NaCl+H2O+CO2��
106 44
x 4.4g
| 106 |
| x |
| 44 |
| 4.4g |
x=10.6g
ԭ�������Na2CO3�������ٷ���=
| 10.6g |
| 50g |
��ԭ�������Na2CO3�������ٷ�����21.2%��
�����������ۺϿ���ʵ�鷽����������������⣬ͬʱҪ��ȷ�������ҳ���Ч���ݽ��м��㣮

��ϰ��ϵ�д�
 ��ս100��Ԫ����Ծ�ϵ�д�
��ս100��Ԫ����Ծ�ϵ�д�
�����Ŀ
�ؿ���Ԫ�ص������������������ɴ�С��˳���ǣ�������
| A���� ̼ �� �� |
| B���� �� �� �� |
| C���� �� �� �� |
| D���� �� �� �� |
ʵ��������ȡ������̼������Ϊ����ʵķ����ǣ�������
| A��ľ̿ȼ�� |
| B������ʯ��ʯ |
| C��ʯ��ʯ��ϡ���ᷴӦ |
| D������ʯ��ϡ���ᷴӦ |
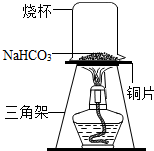 ̼�����Ƴ�����ʳƷ��ҽҩ��ҵ��ij��ѧ��ȤС���ͬѧ��̼�����Ƶ����ȶ��Խ���̽����
̼�����Ƴ�����ʳƷ��ҽҩ��ҵ��ij��ѧ��ȤС���ͬѧ��̼�����Ƶ����ȶ��Խ���̽����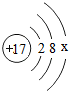 ��x=
��x=