��Ŀ����
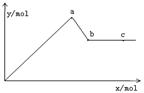
��֪Ba(AlO2)2������ˮ����ͼ��ʾ������Al2(SO4)3��Һ����μ���Ba(OH)2��Һʱ�����ɳ��������ʵ���y�����Ba(OH)2�����ʵ���x�Ĺ�ϵ�������й�������ȷ����
A.a��bʱ���������ʵ�����Al(OH)3��BaSO4��
B.c��dʱ��Һ�����ӵ����ʵ�����![]() ��Ba2+��
��Ba2+��
C.a��dʱ���������ʵ�����BaSO4����С��Al(OH)3
D.d��eʱ��Һ�����ӵ����ʵ�����Ba2+���ܵ���OH-
BD b��ʱ������࣬��ʱAl3+ȫ��ת��ΪAl(OH)3��������ӦΪ��Al2(SO4)3+3Ba(OH)2![]() 2Al(OH)3��+3BaSO4����O��b�����κ�һ����������Ϸ�Ӧ��A����b��d�㷴ӦΪ��Al(OH)3+OH-
2Al(OH)3��+3BaSO4����O��b�����κ�һ����������Ϸ�Ӧ��A����b��d�㷴ӦΪ��Al(OH)3+OH-![]()
![]() +2H2O��d��Al(OH)3��Ӧ��ȫ��a��dʱBaSO4�����ʵ��������Al(OH)3�����ʵ�����dʱ��Һ�к���Ba(AlO2)2��d��eʱ��μ���Ba(OH)2������ʹBa2+��OH-���ʵ������.
+2H2O��d��Al(OH)3��Ӧ��ȫ��a��dʱBaSO4�����ʵ��������Al(OH)3�����ʵ�����dʱ��Һ�к���Ba(AlO2)2��d��eʱ��μ���Ba(OH)2������ʹBa2+��OH-���ʵ������.

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
��֪Ba(AlO2)2������ˮ������1 mol Al2(SO4)3����Һ�м��뺬��b mol Ba(OH)2 (b��6)����Һ�����ó��������ʵ���������Ϊ
| A��5mol | B��3mol | C��b/2mol | D��5b/3mol |
��֪Ba(AlO2)2������ˮ����ͼ��ʾ������A12(SO4)3 0.01mol����Һ����μ���Ba(OH)2��Һʱ�����ɳ��������ʵ���y�����Ba(OH)2�����ʵ���x�Ĺ�ϵ������a��c�ֱ���0b�κ�bd�ε��е㣩�������й�������ȷ����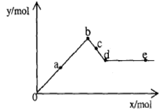
| A��aʱ����������Ϊ3.495 g |
| B��bʱ���������ʵ���Ϊ0.05 mol |
| C��cʱ��Һ��Ba2+���ӵ����ʵ���Ϊ0.005 mol |
| D��eʱ��Һ��AlO2�������ʵ���Ϊ0.01 mol |