��Ŀ����
��20�֣���֪��
��1��2C��ʯīs����O2 (g) =" 2" CO(g) ��H=��222KJ/mol
(g) =" 2" CO(g) ��H=��222KJ/mol
��2��2 H2(g����O2(g)="2" H2O(g) ��H=��484KJ��mol
��3��C��ʯīs����O2��g��=CO2��g�� ��H=��393.5KJ/mo l
l
��4��H2O(g��=H2O��L) ��H=��44KJ/mol
��5��C�����ʯs����O2(g)=CO2(g) ��H=��395.4KJ/mol
�ش��������⣺
��1��C��ʯī����ȼ����Ϊ ��
��2��д����ʾH2��ȼ���ȵ��Ȼ�ѧ����ʽ�� ��
��
��3��д��ʯīת��Ϊ���ʯ���Ȼ�ѧ����ʽ���ɲ�ע�������� ����Ը��ȶ��������� ��
��4����ҵ����H2��һ����Ҫ��Ӧ�ǣ�CO(g)+H2O(g)?CO2 (g)+H2(g)
�ٸ÷�Ӧ�ġ�H= ��
��Ϊ�����CO��ת���ʣ����д�ʩ�ɲ��õ��� ��
��ƽ�ⳣ��K�ı���ʽ��K= ��
��800��ʱ���÷�Ӧ��ƽ�ⳣ��K=1����850��ʱK 1���������������
��800��ʱ������ʼŨ�ȣ�C(CO)="1" mol/L�� C(H2O)=4mol/L�����ƽ��ʱ��C(H2��= ��CO��ת����Ϊ ��

��1��2C��ʯīs����O2
 (g) =" 2" CO(g) ��H=��222KJ/mol
(g) =" 2" CO(g) ��H=��222KJ/mol��2��2 H2(g����O2(g)="2" H2O(g) ��H=��484KJ��mol
��3��C��ʯīs����O2��g��=CO2��g�� ��H=��393.5KJ/mo
 l
l��4��H2O(g��=H2O��L) ��H=��44KJ/mol
��5��C�����ʯs����O2(g)=CO2(g) ��H=��395.4KJ/mol
�ش��������⣺
��1��C��ʯī����ȼ����Ϊ ��
��2��д����ʾH2��ȼ���ȵ��Ȼ�ѧ����ʽ��
 ��
����3��д��ʯīת��Ϊ���ʯ���Ȼ�ѧ����ʽ���ɲ�ע�������� ����Ը��ȶ��������� ��
��4����ҵ����H2��һ����Ҫ��Ӧ�ǣ�CO(g)+H2O(g)?CO2 (g)+H2(g)
�ٸ÷�Ӧ�ġ�H= ��
��Ϊ�����CO��ת���ʣ����д�ʩ�ɲ��õ��� ��
| A�������¶� | B������ѹǿ |
| C��ѡ����ʵĴ��� | D������H2O(g)��Ũ�� |
��800��ʱ���÷�Ӧ��ƽ�ⳣ��K=1����850��ʱK 1���������������
��800��ʱ������ʼŨ�ȣ�C(CO)="1" mol/L�� C(H2O)=4mol/L�����ƽ��ʱ��C(H2��= ��CO��ת����Ϊ ��
��20�֣�
��1��393.5KJ/mol
��2��H2(g)+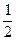 O2(g)=H2O(L) ��H=��286KJ/mol
O2(g)=H2O(L) ��H=��286KJ/mol
��3��C��ʯīs��=C�����ʯs�� ��H="+1.9KJ/mol " ʯī
��4���٣�40.5KJ/mol ��D ��K=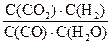 ��<1 ��0.8mol/L, 80%
��<1 ��0.8mol/L, 80%
��1��393.5KJ/mol
��2��H2(g)+
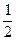 O2(g)=H2O(L) ��H=��286KJ/mol
O2(g)=H2O(L) ��H=��286KJ/mol��3��C��ʯīs��=C�����ʯs�� ��H="+1.9KJ/mol " ʯī
��4���٣�40.5KJ/mol ��D ��K=
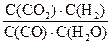 ��<1 ��0.8mol/L, 80%
��<1 ��0.8mol/L, 80%��

��ϰ��ϵ�д�
�����Ŀ
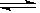 2 SO3 (g); ��H <0, �÷�Ӧ�����Ṥҵ������Ӧ��ȡ�����������ǣ� ��
2 SO3 (g); ��H <0, �÷�Ӧ�����Ṥҵ������Ӧ��ȡ�����������ǣ� ��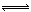 4NO��g����6H2O��g������������ȷ����(���� )��
4NO��g����6H2O��g������������ȷ����(���� )�� qZ(g) ��H��0��m��n��q�������һ�����ܱ�������Ͷ�뷴Ӧ��ﵽƽ�⣬����������ȷ����( )
qZ(g) ��H��0��m��n��q�������һ�����ܱ�������Ͷ�뷴Ӧ��ﵽƽ�⣬����������ȷ����( ) 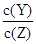 ��ֵ��С
��ֵ��С 2Z(g) ��H<0���ڲ�ͬ�¶ȣ�T1��T2����ѹǿ��P1�� P2���£�����Z�����ʵ���(nz)�뷴Ӧʱ��(t)�Ĺ�ϵ��ͼ��ʾ.�����ж���ȷ����
2Z(g) ��H<0���ڲ�ͬ�¶ȣ�T1��T2����ѹǿ��P1�� P2���£�����Z�����ʵ���(nz)�뷴Ӧʱ��(t)�Ĺ�ϵ��ͼ��ʾ.�����ж���ȷ����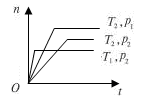
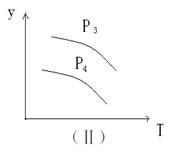
 xC��g��,��������ͼ����ʾ��ϵ���ɴ��ƶ϶�ͼ����ȷ˵����
xC��g��,��������ͼ����ʾ��ϵ���ɴ��ƶ϶�ͼ����ȷ˵����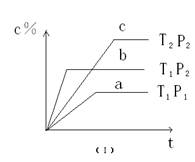
 ƽ����Է�������
ƽ����Է������� CO(g)��H2O(g)��ƽ�ⳣ�����¶ȱ仯���±���
CO(g)��H2O(g)��ƽ�ⳣ�����¶ȱ仯���±��� ���£�
���£� 3Z(��)����֪����1 molX��3molY���ﵽƽ�������a mol Z��
3Z(��)����֪����1 molX��3molY���ﵽƽ�������a mol Z�� X��6 mol Y���ﵽƽ���Z�����ʵ���Ϊ ��
X��6 mol Y���ﵽƽ���Z�����ʵ���Ϊ ��