��Ŀ����
(9��)Ϊ�Ƚ�Fe3+��Cu2+��MnO2��H2O2�ֽⷴӦ�Ĵ�Ч�� ��ij��ѧ�о�С���ͬѧ�ֱ��������ͼ�ס��ҡ�����ʾ��ʵ�顣��ش�������⣺
��ij��ѧ�о�С���ͬѧ�ֱ��������ͼ�ס��ҡ�����ʾ��ʵ�顣��ش�������⣺

��1��ͬѧX�۲������֧�Թܲ������ݵĿ������ɴ˵ó�Fe3+��Cu2+��H2O2�ֽ�Ĵ�Ч���������________(���������������)��������_______________________��
��2��ͬѧY������װ�ã��ⶨ��ͬʱ���ڲ��������������Ӷ�̽��Fe3+��Cu2+��H2O2�ֽ�Ĵ�Ч�����Իش��������⣺
������A��������____ ____��
____��
�ڳ��˱�Ҫ���Լ���ͼ����ʾ�������⣬����һ������ȱ�ٵ�ʵ����Ʒ��___________��
��ʵ��ʱ��װ��װ���ң��ر�A�Ļ�������ע����������������һ�Ρ���һ������ʵ��Ŀ����_______________________��
�ܲⶨ�������ʱ���Ƿ���Ҫ�����ų���װ���ڵĿ���________(���Ҫ������Ҫ��)��
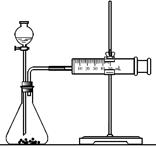
��3��ͬѧZ���ñ�װ��̽��MnO2��H2O2�ֽ�Ĵ�Ч������50 mL ˫��ˮһ���Լ���ʢ��0.10 mol MnO2��ĩ����ƿ�У���ñ�״�����������ܶ�����������[V(������)]��
ʱ��(t/min)�Ĺ�ϵ��ͼ��ʾ��

��ʵ��ʱ�ų�������������________mL��
��b________(����ڡ�����С�ڡ����ڡ�)90 mL��
��ͬѧZ����̽��MnO2��H2O2�ֽ����ʵ�Ӱ�������
���ɵó�________��H2O2�ֽ����ʵ�Ӱ�������
 ��ij��ѧ�о�С���ͬѧ�ֱ��������ͼ�ס��ҡ�����ʾ��ʵ�顣��ش�������⣺
��ij��ѧ�о�С���ͬѧ�ֱ��������ͼ�ס��ҡ�����ʾ��ʵ�顣��ش�������⣺
��1��ͬѧX�۲������֧�Թܲ������ݵĿ������ɴ˵ó�Fe3+��Cu2+��H2O2�ֽ�Ĵ�Ч���������________(���������������)��������_______________________��
��2��ͬѧY������װ�ã��ⶨ��ͬʱ���ڲ��������������Ӷ�̽��Fe3+��Cu2+��H2O2�ֽ�Ĵ�Ч�����Իش��������⣺
������A��������____
 ____��
____���ڳ��˱�Ҫ���Լ���ͼ����ʾ�������⣬����һ������ȱ�ٵ�ʵ����Ʒ��___________��
��ʵ��ʱ��װ��װ���ң��ر�A�Ļ�������ע����������������һ�Ρ���һ������ʵ��Ŀ����_______________________��
�ܲⶨ�������ʱ���Ƿ���Ҫ�����ų���װ���ڵĿ���________(���Ҫ������Ҫ��)��
��3��ͬѧZ���ñ�װ��̽��MnO2��H2O2�ֽ�Ĵ�Ч������50 mL ˫��ˮһ���Լ���ʢ��0.10 mol MnO2��ĩ����ƿ�У���ñ�״�����������ܶ�����������[V(������)]��
ʱ��(t/min)�Ĺ�ϵ��ͼ��ʾ��

��ʵ��ʱ�ų�������������________mL��
��b________(����ڡ�����С�ڡ����ڡ�)90 mL��
��ͬѧZ����̽��MnO2��H2O2�ֽ����ʵ�Ӱ�������
���ɵó�________��H2O2�ֽ����ʵ�Ӱ�������

��

��ϰ��ϵ�д�
 �ܿ�����ĩ��̾�ϵ�д�
�ܿ�����ĩ��̾�ϵ�д�
�����Ŀ
 T/K
T/K ��״��ͬ
��״��ͬ ������ͬ��
������ͬ��
 Ӧ������������ѧ����֪ʶ��������Ӧ���������ԭ����________________ __________________��������һ��ʱ���Ӧ�����ּ�����������ʼ�����ԭ����_______________________________��
Ӧ������������ѧ����֪ʶ��������Ӧ���������ԭ����________________ __________________��������һ��ʱ���Ӧ�����ּ�����������ʼ�����ԭ����_______________________________�� CH3COCH2I��H����I����25��ʱ���÷�Ӧ�����������о��鹫ʽ������
CH3COCH2I��H����I����25��ʱ���÷�Ӧ�����������о��鹫ʽ������ 2SO3�����2 min��SO2������12 mol�½�Ϊ4 mol��������˵����ȷ����
2SO3�����2 min��SO2������12 mol�½�Ϊ4 mol��������˵����ȷ����