��Ŀ����
20��������N2H4��������������һ����Ҫ�Ļ��ȼ�ϣ�N2H4��N2O4��Ӧ�ܷų��������ȣ���1����֪��2NO2��g��?N2O4��g����H=-57.20kJ•mol-1��һ���¶��£����ܱ������д˷�Ӧ�ﵽƽ�⣮ ������������ʱ�����д�ʩ����ʹNO2ת������ߵ���CD
A������NO2��Ũ�� B�������¶� C������NO2��Ũ�� D�������¶�
��2��25��ʱ��1.00gN2H4��l��������N2O4��l����ȫ��Ӧ����N2��g����H2O��l�����ų�19.14kJ����������Ӧ2N2H4��l��+N2O4��l���T3N2��g��+4H2O��l���ġ�H=-1224.96 kJ•mol-1��
��3��17�桢1.01��105Pa���ܱ�������N2O4��NO2�Ļ������ﵽƽ��ʱ��c��NO2��=0.0300mol•L-1��c��N2O4��=0.0120mol•L-1�����㷴Ӧ2NO2��g��?N2O4��g����ƽ�ⳣ��K=13.33��
��4����������ˮ�������백���Ƶ�����õ��뷽��ʽ��ʾ�µ�ˮ��Һ�Լ��Ե�ԭ��N2H4+H2O
 N2H+5+OH-��
N2H+5+OH-����5����֪����ͬ������N2H4•H2O�ĵ���̶ȴ���N2H5C1��ˮ��̶ȣ������£�����0.2mol/L N2H4•H2O��Һ��0.1mol/L HCl��Һ�������ϣ�����Һ��N2H5+��Cl-��OH-��H+����Ũ���ɴ�С��˳��Ϊ��c��N2H+5����c��Cl-����c��OH-����c��H+������
��6����-����ȼ�ϵ����һ�ּ���ȼ�ϵ�أ��������Һ��20%��30%��KOH��Һ����-����ȼ�ϵ�طŵ�ʱ�������ĵ缫��Ӧʽ��N2H4+4OH--4e-=N2+4H2O����ع���һ��ʱ��������Һ��pH����С�����������С���������䡱����
���� ��1��A������NO2��Ũ�ȣ���ЧΪ����ѹǿ��
B�������¶ȣ�ƽ������ȷ�Ӧ�����ƶ���
C������NO2��Ũ�ȣ���ЧΪ����ѹǿ��
D�������¶ȣ�ƽ�������ȷ�Ӧ�����ƶ���
��2������2molN2H4��Ӧ�ų������������ȷ�Ӧ���ʱ��H��0��
��3������ƽ�ⳣ������ʽK=$\frac{c��{N}_{2}{O}_{4}��}{{c}^{2}��N{O}_{2}��}$���㣻
��4��N2H4���H2O�����������ʹ��Һ�ʼ��ԣ�
��5����0.2mol/L N2H4•H2O��Һ��0.1mol/L HCl��Һ�������ϣ��õ�������N2H4•H2O��N2H5C1������ͬ������N2H4•H2O�ĵ���̶ȴ���N2H5C1��ˮ��̶ȣ�����Һ�Լ��ԣ���c��OH-����c��H+����
��6����--����ȼ�ϵ�أ���������������Ӧ��N2H4�ڷ���ʧȥ���Ӽ������������ɵ�����ˮ��������N2H4ȼ�����ɵ�����ˮ����Һ�����������ӵ�Ũ�ȱ�С��
��� �⣺��1��A������NO2��Ũ�ȣ���ЧΪ����ѹǿ��ƽ�������ƶ���NO2ת��������A��ѡ��
B������ӦΪ���ȷ�Ӧ�������¶ȣ�ƽ��������Ӧ�����ƶ���NO2ת��������B��ѡ��
C������NO2��Ũ�ȣ���ЧΪ����ѹǿ��ƽ�����淴Ӧ�����ƶ���NO2ת���ʼ�С����Cѡ��
D������ӦΪ���ȷ�Ӧ�������¶ȣ�ƽ�����淴Ӧ�ƶ���NO2ת���ʼ�С����Dѡ��
��ѡ��CD��
��2��2molN2H4��Ӧ�ų�������Ϊ19.14kJ��$\frac{2mol��32g/mol}{1g}$=1224.96kJ����Ӧ2N2H4��l��+N2O4��l���T3N2��g��+4H2O��l���ġ�H=-1224.96kJ•mol-1
�ʴ�Ϊ��-1224.96��
��3���������ﵽƽ��ʱ��c��NO2��=0.0300mol•L-1��c��N2O4��=0.0120mol•L-1����Ӧ2NO2��g��?N2O4��g����ƽ�ⳣ��K=$\frac{c��{N}_{2}{O}_{4}��}{{c}^{2}��N{O}_{2}��}$=$\frac{0.012}{0.0{3}^{2}}$=13.33��
�ʴ�Ϊ��13.33��
��4����������ˮ�������백���Ƶ�������������OH-�������ӣ����뷽��ʽΪN2H4+H2O N2H+5+OH-��
N2H+5+OH-��
�ʴ�Ϊ��N2H4+H2O N2H+5+OH-��
N2H+5+OH-��
��5����0.2mol/L N2H4•H2O��Һ��0.1mol/L HCl��Һ�������ϣ��õ�������N2H4•H2O��N2H5C1������ͬ������N2H4•H2O�ĵ���̶ȴ���N2H5C1��ˮ��̶ȣ���c��N2H+5����c��Cl-��������Һ�Լ��ԣ���c��OH-����c��H+�������롢ˮ��ij̶ȶ���������c��N2H+5����c��Cl-����c��OH-����c��H+����
�ʴ�Ϊ��c��N2H+5����c��Cl-����c��OH-����c��H+����
��6����--����ȼ�ϵ�أ���������������Ӧ��N2H4�ڷ���ʧȥ���Ӽ������������ɵ�����ˮ�������缫��ӦʽΪ��N2H4+4OH--4e-=N2+4H2O���õ�ط�ӦΪN2H4+O2=N2+2H2O����Ӧ����ˮ����Һ�����������ӵ�Ũ�ȱ�С���������Һ��pH����С��
�ʴ�Ϊ��N2H4+4OH--4e-=N2+4H2O����С��
���� ���⿼�黯ѧƽ���ƶ�����Ӧ�ȼ��㡢ƽ�ⳣ�����㡢����Ũ�ȴ�С�Ƚϡ�ԭ��صȣ��Ƕ�ѧ���ۺ������Ŀ��飬��Ҫѧ���߱���ʵ�Ļ������Ѷ��еȣ�

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�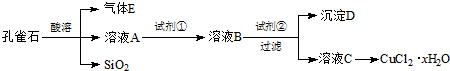
��֪���йؽ������Ӵӿ�ʼ������������ȫʱ��Һ��pH��
| �������� pH | Fe3+ | Cu2+ | Fe2+ |
| ��ʼ���� | 2.7 | 5.4 | 8.1 |
| ������ȫ | 3.7 | 6.9 | 9.6 |
A��Cu����B��Cl2�� C��NH3•H2O����D��NaOH E��H2O2����F��KMnO4
��2�������Լ��ڵ�Ŀ���ǵ���pH��a��a�ķ�Χ��3.7��5.4��д���Լ��ڵ�һ�ֿ��ܵĻ�ѧʽCuO��Cu��OH��2������ҺC���CuCl2•xH2O������4������ʵ���������4������ʵ���������������Ũ������ȴ�ᾧ�����ˡ�����ˮ�Ҵ�ϴ�ӵȲ�����ʹ����ˮ�Ҵ�����ˮ����ϴ�ӵ���Ҫԭ���Ƿ�ֹCuCl2•xH2O�����ܽ���ʧ��
��3��Ϊ�˲ⶨ�Ƶõ��Ȼ�ͭ���壨CuCl2•xH2O����xֵ����ͬѧ�������������ʵ�鷽����
�ٳ�ȡһ�������ľ������ʹ��ʧȥ�ᾧˮ������������ˮCuCl2���������÷������ڵ�������CuCl2•xH2O�������ʱ�ᷢ��ˮ�⣮
�ڳ�ȡag���塢������������������Һ�����ˡ�ϴ�ӡ����ȳ������������ټ���Ϊֹ���������ù��������Ϊbg����x=$\frac{80a-135b}{18b}$���ú�a��b�Ĵ���ʽ��ʾ����
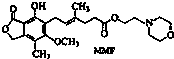 ù��������MMF����������ֲ������ϸ����ֳ��õ�ҩ��ṹ��ʽ���£����й���MMF˵���в���ȷ���ǣ�������
ù��������MMF����������ֲ������ϸ����ֳ��õ�ҩ��ṹ��ʽ���£����й���MMF˵���в���ȷ���ǣ�������| A�� | MMF�ȿ�����Br2��CCl4��Һ�����ӳɷ�Ӧ���ֿ����ڹ�������Br2��Ӧ����ȡ�� | |
| B�� | 1molMMF���뺬3molNaOH��ˮ��Һ��ȫ��Ӧ | |
| C�� | MMF�����Դ����⣬�ֿ���ʹ���Ը��������Һ��ɫ | |
| D�� | MMF�ȿ�����FeCl3��Һ������ɫ��Ӧ���ֿ�����NaHCO3��Һ��Ӧ�ų�CO2 |
| A�� | �ʱ���ָ1mol������ȫ�μӷ�Ӧʱ�������仯 | |
| B�� | �к͵ζ�ʵ��ʱ���ô���Һ��ϴ��ƿ | |
| C�� | ��֪��2HI��g��?H2��g��+I2��g����H=8.8kJ•mol-1�������ܱ������г���1molHI��g������ֽ���������յ�����Ϊ4.4kJ | |
| D�� | ���ڷ�Ӧ3H2��g��+N2��g��?2NH3��g������ʹ�ô�����v����v��ͬ�ȳ̶ȼӿ� |
| A�� | CaO | B�� | ����� | C�� | NaOH��Һ | D�� | Ũ���� |
| A�� | �������Ħ������ 71 g•mol-1 | |
| B�� | ��������2.24 L�����庬������Ϊ1.7 NA | |
| C�� | ��1mol������ͨ��������NaOH�з�Ӧ��ת�Ƶ�����Ϊ NA | |
| D�� | �������ˮ��Һ����ǿ�����ԣ���ɱ��������Ư��ijЩ���� |
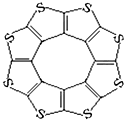 ������³�Ƚ���ѧ�о����Ľ���ADF������һ�����ͻ�ϩ�ഢ����ϣ�C16S8�������о��������۽Ƕ�֤�����ַ����е�ԭ�Ӷ�����ͬһƽ���ϣ��ṹ��ͼ��ʾ����ÿ��ƽ�������������ɴ���10��H2���ӣ�
������³�Ƚ���ѧ�о����Ľ���ADF������һ�����ͻ�ϩ�ഢ����ϣ�C16S8�������о��������۽Ƕ�֤�����ַ����е�ԭ�Ӷ�����ͬһƽ���ϣ��ṹ��ͼ��ʾ����ÿ��ƽ�������������ɴ���10��H2���ӣ���Ԫ�ص縺�Դ�С��ϵ�ǣ�C��S�����������=����������
�ڷ�����Cԭ�ӵ��ӻ��������Ϊsp2��
���йؼ����������£�
| C-S | C=S | C16S8��̼��� | |
| ����/pm | 181 | 155 | 176 |
��C16S8��H2������������Ƿ��»�����
| A�� | ���� | B�� | ��ϩ | C�� | ������ | D�� | �������� |