��Ŀ����
������й���ļ������Σ���������������ظ���Ԫ��Ϊ������NaCl����ṹ����ͼ��ʾ����֪FexO���徧���ṹΪNaCl�ͣ����ھ���ȱ�ݣ�xֵС��1����֪FexO�����ܶ�Ϊ��=5.71 g��cm-3�������߳�Ϊ4.28��10-10 m��
��1��FexO��xֵ(��ȷ��0.01)Ϊ____________��
��2�������е�Fen+�ֱ�ΪFe2+��Fe3+,��Fe2+��Fe3+�������У�Fe2+��ռ��������С����ʾ����ȷ��0.001��Ϊ____________________��
��3���˾��廯ѧʽΪ______________��
��4����ij��Fe2+(��Fe3+)��������ҵȾ����O2-Χ�ɵĿռ伸����״��___________��
��5���ھ����У���Ԫ�ص����Ӽ���̾���Ϊ____________m��
����������NaCl����ṹ��1��NaCl��������8��С�����塲����ͼ(1)�����ɵġ�ÿ��С�����庬12��NaCl��ͬ����FexO�����У�ÿ��С�����庬12��FexO�����������С�����ͼ(2)����Fe��O������֣�x=1��

��1��ÿ��С�����������Ϊ��m=��V
=5.71 g��cm-3��(![]() )3
)3
=5.60��10-23 g��
M��FexO��=2m��NA=2��5.60��10-23 g��6.02��1023mol-1=67.4 g��mol-1��
56.0 g��mol-1��x+16.0 g��mol-1��1=67.4 g��mol-1
x=0.92��
��2����1 mol Fe0.92O��N(Fe2+)=y mol,
��N(Fe3+)=(0.92-y)mol�����ݻ������и�Ԫ���������ϼ۴�����Ϊ���ԭ��2��y mol+3��(0.92-y mol)+(-2)��1 mol=0,y=0.76��
��Fe2+��ռ����Ϊ0.76/0.92=0.826��
��3������Fe2+Ϊ0.76,��Fe3+Ϊ0.92-0.76=0.16���ʻ�ѧʽΪ��![]() ��
��
��4��Fe�ھ�������ռ��϶�ļ�����״Ϊ�������塣��ͼ(3)��ʾ��

ͼ(3)
��5����ͼ(1)����Ԫ�����Ӽ�ľ���
r=![]() =1.41��4.28��10-10 m��
=1.41��4.28��10-10 m��![]() =3.02��10-10 m��
=3.02��10-10 m��
�𰸣���1��0.92
��2��0.826��
��3��![]()
��4����������
��5��3.02��10-10



 2s
2s 3s
3s 4s
4s �����Ԫ�ػ�̬ԭ�ӵĵ����Ų�ʽΪ ��������������Ӧ��ˮ����Ļ�ѧʽ��
�����Ԫ�ػ�̬ԭ�ӵĵ����Ų�ʽΪ ��������������Ӧ��ˮ����Ļ�ѧʽ�� 
 2s
2s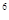 3s
3s 4s
4s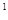 �����Ԫ�ػ�̬ԭ�ӵĵ����Ų�ʽΪ
��������������Ӧ��ˮ����Ļ�ѧʽ��
�����Ԫ�ػ�̬ԭ�ӵĵ����Ų�ʽΪ
��������������Ӧ��ˮ����Ļ�ѧʽ��