��Ŀ����
ͭ���仯�������������������й㷺��Ӧ�á�
(1)ͭ�ɲ������·����Ʊ���
����ͭ��Cu2S��O2 2Cu��SO2
2Cu��SO2
ʪ����ͭ��CuSO4��Fe=FeSO4��Cu
�������ַ����У�ͭԪ�ؾ���________(���������ԭ��)��ͭ���ʡ�
(2)ӡˢ��·����ʹ�õ�ͭ��Ҫ�������á�
����һ����FeCl3��Һ����ӡˢ��·���Ʊ�CuCl2��2H2O��ʵ����ģ����չ������£�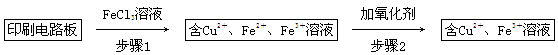

��֤������1����FeCl3��Һ�����ķ�����_________________________________��
�ڲ���2�����ӵ������������˵���______________________________________��
A��HNO3 B��H2O2 C��KMnO4
�۲���3��Ŀ����ʹ��Һ��pH���ߵ�4.2����ʱFe3����ȫ��������ѡ�õġ��Լ�1����________��(д��һ�ּ���)
������Ũ��CuCl2��Һʱ��Ҫ�μ�Ũ���ᣬĿ����________(�û�ѧ����ʽ����ϼ�Ҫ������˵��)���پ���ȴ���ᾧ�����ˣ��õ�CuCl2��2H2O��
����������H2O2��ϡ���Ṳͬ����ӡˢ��·���Ʊ�����ͭʱ�����Ȼ�ѧ����ʽ�ǣ�
Cu(s)��H2O2(l)��H2SO4(aq)=CuSO4(aq)��2H2O(l)����H1����320 kJ��mol��1
��֪��2H2O2(l)=2H2O(l)��O2(g)��H2����196 kJ��mol��1
H2(g)��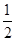 O2(g)=H2O(l)����H3����286 kJ��mol��1
O2(g)=H2O(l)����H3����286 kJ��mol��1
��ӦCu(s)��H2SO4(aq)=CuSO4(aq)��H2(g)�Ħ�H��________��
(3)��ʵ�ַ�ӦCu��H2SO4=CuSO4��H2����������Ϊ��ʵ�ָ�ת����װ���е������ڣ�����缫����(�Cu����C��)��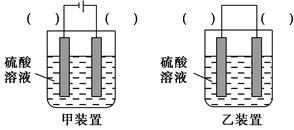
(1)��ԭ��(2)����һ����ȡ������ַ�Ӧ�����Һ���Թ��У��μ�KSCN��Һ������Һ��Ϊ��ɫ��֤������FeCl3��Һ��������B����CuO��Cu(OH)2(�𰸺�������)����CuCl2����Һ���ܷ���ˮ�ⷴӦ��CuCl2��2H2O??Cu(OH)2��2HCl���μ�Ũ���ᣬ����������ˮ��
����������64 kJ��mol��1
(3)
����

 ��ĩ���䵥Ԫ�����ิϰ��ϵ�д�
��ĩ���䵥Ԫ�����ิϰ��ϵ�д�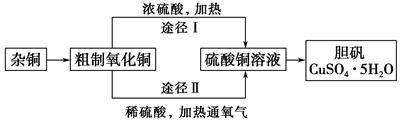




 ____CuAlO2��________����
____CuAlO2��________����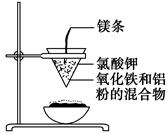


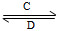 B(��ˮ��Һ�н���)
B(��ˮ��Һ�н���)
