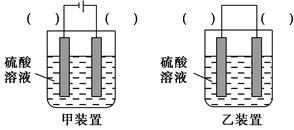
��Ŀ����
ij�����о�С�飬�ú��н϶����ʵ�ͭ�ۣ�ͨ����ͬ�Ļ�ѧ��Ӧ��ȡ����������Ƶ�ʵ�����Ϊ��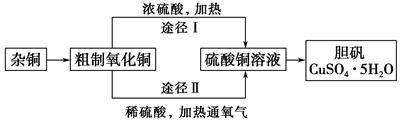
(1)��ͭ�к��д������л���ɲ������յķ�����ȥ�л������ʱ������������ ��(���������������ı�����룬��ͬ)��ȡ������Ӧʹ�� �����պ������Ӧ���� �ϣ�����ֱ�ӷ��������ϡ�
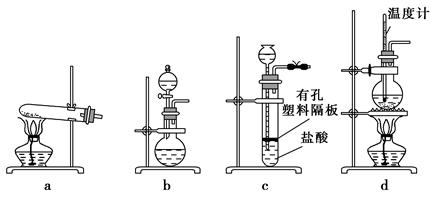
ʵ������������
a������bʯ������c�����ǣ�d������e����ǯ��f�Թܼ�
(2)��ͭ�����պ�õ��IJ���������ͭ������ͭ�Ļ������պ�������ͭ�Ŀ���ԭ���� ��
a�����չ����в�������ͭ����ԭ
b�����ղ����ͭδ����ȫ����
c������ͭ�ڼ��ȹ����зֽ�����ͭ
d����������ͭ������������
(3)ͨ��;����ʵ���ô�������ͭ��ȡ������������е�ʵ��������裺���ܡ�����ͨ���������ˡ� ����ȴ�ᾧ�� ����Ȼ���
(4)�ɴ�������ͭͨ������;����ȡ��������;������ȣ�;���������Ե������ŵ��� �� ��
(5)�ڲⶨ���õ���(CuSO4��xH2O)�нᾧˮxֵ��ʵ������У������������ٽ��� �Ρ�
(6)���ⶨ���xֵƫ�ߣ����ܵ�ԭ���� (����ĸ���)��
a�������¶ȹ���
b����������Ŀ����ϴ�
c�����Ⱥ���ڿ�������ȴ
d���������岿�ַ绯
(1)c��e��b
(2)a��b
(3)����������
(4)������������;�������������١�;���������Ⱦ����������
(5)4
(6)a
����

�������й����ʵ�ת����ϵͼ(��Щ������ʡ��)��
��AΪ���ʣ�E�ڳ�����ΪҺ�壬C����Է�������Ϊ78����ش��������⣺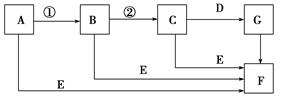
(1)����A��ԭ�ӽṹʾ��ͼ________��F�ĵ���ʽ��________��
(2)�����C���ʽṹ�����ʵ��ƶ��У�����ȷ����________��
| A�������ڿ����л��ɰ�ɫ |
| B������ǿ������ |
| C�������д������Ӽ����ۼ� |
| D����ʪ�����ɫʯ����ֻֽ��ʹ�����ɫ |
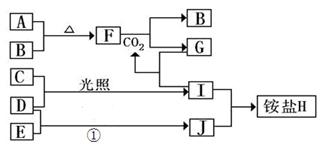
 2Fe(s)��3CO(g�� ��H����492.7kJ/mol
2Fe(s)��3CO(g�� ��H����492.7kJ/mol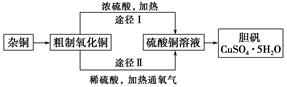
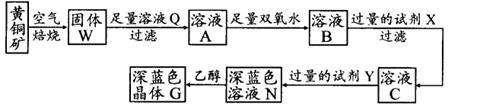
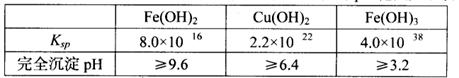
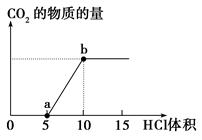
 2Cu��SO2
2Cu��SO2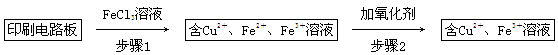

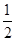 O2(g)=H2O(l)����H3����286 kJ��mol��1
O2(g)=H2O(l)����H3����286 kJ��mol��1