��Ŀ����
��1mol I2 ��g����2 mol H2����ij�ݻ�Ϊ2 L���ܱ������У���һ���¶��·�����Ӧ��I2 ��g��+H2 ��g�� ![]() 2HI��g�����÷�Ӧ����Ӧ���ȣ������ﵽƽ�⡣HI���������
2HI��g�����÷�Ӧ����Ӧ���ȣ������ﵽƽ�⡣HI���������![]() ��HI����ʱ��ı仯��ͼ�����ߣ�����ʾ��
��HI����ʱ��ı仯��ͼ�����ߣ�����ʾ��

��1���ﵽƽ��ʱ��I2 ��g�������ʵ���Ũ��Ϊ_________mol/L��
��2�����ı䷴Ӧ�������ڼ�������![]() ��HI���ı仯��ͼ�����ߣ�����ʾ���������������______________ ������������������ţ�
��HI���ı仯��ͼ�����ߣ�����ʾ���������������______________ ������������������ţ�
�ٺ��������£������¶� �ں��������£������¶�
�ۺ��������£���С��Ӧ��������� �ܺ��������£�����Ӧ���������
�ݺ��º��������£������ʵ�����
��3���������ݻ��ȶ����䣬����һ�ݻ�ҲΪ2 L���ܱ������м���a mol I2 ��g����b mol H2��c mol HI��a��b��c������0����������Ӧ�ﵽƽ��ʱ��HI�����������Ϊ0.60����a��b��c�Ĺ�ϵ��______________��
��1��0.05 ��2���ۢ� ��3��2b-4a=c

��ϰ��ϵ�д�
 ȫ�ŵ�����Ԫ�ƻ�ϵ�д�
ȫ�ŵ�����Ԫ�ƻ�ϵ�д�
�����Ŀ
 ��1mol I2��g�� ��2mol H2����2L�ܱ������У���һ���¶��·�����Ӧ��I2��g��+H2��g��?2HI��g������H��0������ƽ�⣮HI���������w��HI����ʱ��仯��ͼ���ߣ�����ʾ��
��1mol I2��g�� ��2mol H2����2L�ܱ������У���һ���¶��·�����Ӧ��I2��g��+H2��g��?2HI��g������H��0������ƽ�⣮HI���������w��HI����ʱ��仯��ͼ���ߣ�����ʾ�� ��1mol I2��g����2mol H2����5L�ܱ������У���һ���¶��·�����Ӧ��I2��g��+H2��g��?2HI��g����H��0�����ﵽƽ�⣮HI���������w��HI����ʱ��仯�����ߣ�����ʾ��
��1mol I2��g����2mol H2����5L�ܱ������У���һ���¶��·�����Ӧ��I2��g��+H2��g��?2HI��g����H��0�����ﵽƽ�⣮HI���������w��HI����ʱ��仯�����ߣ�����ʾ��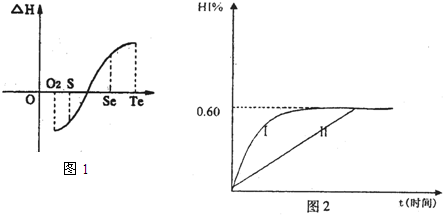

 ��
�� ��2H2O
��2H2O Al��OH��3�� ��NH3��H2O�����е����ʵ�����
Al��OH��3�� ��NH3��H2O�����е����ʵ����� 2HI��g������H<0�� ����ƽ�⡣HI���������HI����ʱ�ʱ仯������ͼ2��ʾ��
2HI��g������H<0�� ����ƽ�⡣HI���������HI����ʱ�ʱ仯������ͼ2��ʾ��
