��Ŀ����
����Һ�У���ӦA +2B C�ֱ������ֲ�ͬʵ�������½��У����ǵ���ʼŨ�Ⱦ�Ϊc( A)=0.100 mol/L��c(B) =0.200 mol/L��c(C) =0 mol/L����Ӧ��A��Ũ����ʱ��ı仯����ͼ��ʾ��
C�ֱ������ֲ�ͬʵ�������½��У����ǵ���ʼŨ�Ⱦ�Ϊc( A)=0.100 mol/L��c(B) =0.200 mol/L��c(C) =0 mol/L����Ӧ��A��Ũ����ʱ��ı仯����ͼ��ʾ��
 C�ֱ������ֲ�ͬʵ�������½��У����ǵ���ʼŨ�Ⱦ�Ϊc( A)=0.100 mol/L��c(B) =0.200 mol/L��c(C) =0 mol/L����Ӧ��A��Ũ����ʱ��ı仯����ͼ��ʾ��
C�ֱ������ֲ�ͬʵ�������½��У����ǵ���ʼŨ�Ⱦ�Ϊc( A)=0.100 mol/L��c(B) =0.200 mol/L��c(C) =0 mol/L����Ӧ��A��Ũ����ʱ��ı仯����ͼ��ʾ�� 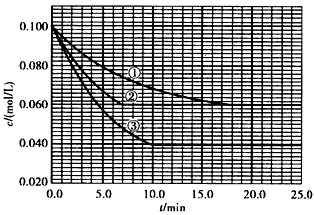
��ش��������⣺
��1����ٱȽϣ��ں͢۷ֱ���ı�һ�ַ�Ӧ���������ı���������жϵ������ǣ�
��_________________________________________________��
��_________________________________________________��
��2��ʵ���ƽ��ʱB��ת����Ϊ________ ��ʵ���ƽ��ʱC��Ũ��Ϊ________��
��3���÷�Ӧ�ġ�H_____ 0�����ж�������_______
��4���÷�Ӧ���е�4.0 minʱ��ƽ����Ӧ���ʣ�
ʵ��ڣ�VB=____________��
ʵ��ۣ�VC=____________��
��1����ٱȽϣ��ں͢۷ֱ���ı�һ�ַ�Ӧ���������ı���������жϵ������ǣ�
��_________________________________________________��
��_________________________________________________��
��2��ʵ���ƽ��ʱB��ת����Ϊ________ ��ʵ���ƽ��ʱC��Ũ��Ϊ________��
��3���÷�Ӧ�ġ�H_____ 0�����ж�������_______
��4���÷�Ӧ���е�4.0 minʱ��ƽ����Ӧ���ʣ�
ʵ��ڣ�VB=____________��
ʵ��ۣ�VC=____________��
��1���Ӵ������ﵽƽ���ʱ�����̣�ƽ��ʱA��Ũ��δ�䣻�¶����ߣ��ﵽƽ���ʱ�����̣�ƽ��ʱA��Ũ�ȼ�С
��2��40%(��0. 4)��0.060 mol/L
��3��> ���¶����ߣ�ƽ��������Ӧ�����ƶ����ʸ÷�Ӧ�����ȷ�Ӧ
��4��0.014mol/(L��min)��0.008mol/(L��min)��
��2��40%(��0. 4)��0.060 mol/L
��3��> ���¶����ߣ�ƽ��������Ӧ�����ƶ����ʸ÷�Ӧ�����ȷ�Ӧ
��4��0.014mol/(L��min)��0.008mol/(L��min)��

��ϰ��ϵ�д�
�����Ŀ
 ��2010?����������Һ�У���ӦA+2B?C�ֱ������ֲ�ͬʵ�������½��У����ǵ���ʼŨ�Ⱦ�Ϊc��A��=0.100mol/L��c��B��=0.200mol/L �� c��C��=0mol/L����Ӧ��A��Ũ����ʱ��ı仯��ͼ��ʾ��
��2010?����������Һ�У���ӦA+2B?C�ֱ������ֲ�ͬʵ�������½��У����ǵ���ʼŨ�Ⱦ�Ϊc��A��=0.100mol/L��c��B��=0.200mol/L �� c��C��=0mol/L����Ӧ��A��Ũ����ʱ��ı仯��ͼ��ʾ��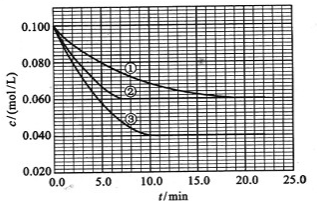 ��2013?Ϋ��ģ�⣩����Һ�У���ӦA+2B?C�ֱ������ֲ�ͬʵ�������½��У����ǵ���ʼŨ�Ⱦ�Ϊc��A��=0.1mol/L��c��B��=0.2mol/L��c��C��=0mol/L����Ӧ��A��Ũ����ʱ��ı仯��ͼ��ʾ��
��2013?Ϋ��ģ�⣩����Һ�У���ӦA+2B?C�ֱ������ֲ�ͬʵ�������½��У����ǵ���ʼŨ�Ⱦ�Ϊc��A��=0.1mol/L��c��B��=0.2mol/L��c��C��=0mol/L����Ӧ��A��Ũ����ʱ��ı仯��ͼ��ʾ�� ����Һ�У���ӦA+2B?C�ֱ������ֲ�ͬʵ�������½��У����ǵ���ʼŨ�Ⱦ�Ϊ c��A��=1.0mol/L��c��B��=2.0mol/L��c��C��=0mol/L����Ӧ��A��Ũ����ʱ��ı仯��ͼ��ʾ����ش��������⣺
����Һ�У���ӦA+2B?C�ֱ������ֲ�ͬʵ�������½��У����ǵ���ʼŨ�Ⱦ�Ϊ c��A��=1.0mol/L��c��B��=2.0mol/L��c��C��=0mol/L����Ӧ��A��Ũ����ʱ��ı仯��ͼ��ʾ����ش��������⣺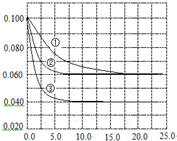 ��2011?��һģ������Һ�У���ӦA+2B?C�ֱ������ֲ�ͬʵ�������½��У����ǵ���ʼŨ�Ⱦ�Ϊc��A��=0.100mol?L-1c��B��=0.200mol?L-1 c��C��=0mol?L-1����Ӧ��A��Ũ����ʱ��ı仯��ͼ��ʾ������˵����ȷ���ǣ�������
��2011?��һģ������Һ�У���ӦA+2B?C�ֱ������ֲ�ͬʵ�������½��У����ǵ���ʼŨ�Ⱦ�Ϊc��A��=0.100mol?L-1c��B��=0.200mol?L-1 c��C��=0mol?L-1����Ӧ��A��Ũ����ʱ��ı仯��ͼ��ʾ������˵����ȷ���ǣ�������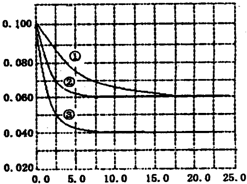 ��2011?֣�ݶ�ģ������Һ�У���ӦA+2B?C�ֱ������ֲ�ͬʵ�������½��У����ǵ���ʼ״̬��Ϊc��A��=0.100mol?L-1��c��B��=0.200mol?L-1��c��C��=0mol?L-1����������
��2011?֣�ݶ�ģ������Һ�У���ӦA+2B?C�ֱ������ֲ�ͬʵ�������½��У����ǵ���ʼ״̬��Ϊc��A��=0.100mol?L-1��c��B��=0.200mol?L-1��c��C��=0mol?L-1����������