��Ŀ����
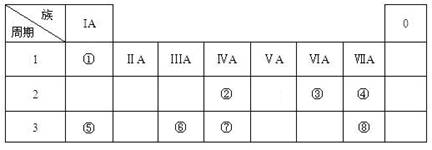
(16�֣�����ΪԪ�����ڱ��е�һ���֣���ע�����������û�ѧ����ش��������⣺
 | ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0 |
| 2 | | | | | | �� | �� | |
| 3 | �� | �� | �� | | | | | �� |
| 4 | �� | �� | | | | | | |
�Ƣ٢ڢݵ�����������Ӧˮ����ļ�����ǿ������˳��Ϊ ��
�Ǣڢۢ����γɵļ����Ӱ뾶�ɴ�С��˳��Ϊ ��
�Ȣ��⻯�ﳣ������Ԫ�آڵĹ������ﷴӦ�����ӷ���ʽΪ ��
�ɢٺ͢��γɵĵ���ɫ����ĵ���ʽΪ ��������ѧ��������Ϊ������Ӽ������ۼ����� ��
�ʢٺ͢ݵ�����������Ӧˮ�������Ӧ�����ӷ���ʽΪ ��
(16�֣���Ar��F����KOH��NaOH��Al��OH)3����K+��Ca2+��Mg2+�� �E
��2H2O+2Na2O2=4Na+ +4OH- +O2����
O�E�E�E�E::
 �����Ӽ����ۼ���
�����Ӽ����ۼ�����OH- +Al��OH)3 =AlO2- +2H2O
����

 ����ѧ��ţ��Ӣ��ϵ�д�
����ѧ��ţ��Ӣ��ϵ�д�(16�֣�����ΪԪ�����ڱ��е�һ���֣���ע�����������û�ѧ����ش��������⣺
|
| ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0 |
| 2 |
|
|
|
|
| �� | �� |
|
| 3 | �� | �� | �� |
|
|
|
| �� |
| 4 | �� | �� |
|
|
|
|
|
|
�� 8��Ԫ���У���ѧ��������õ��� ���ǽ�������ǿ���� ��
�� �٢ڢݵ�����������Ӧˮ����ļ�����ǿ������˳��Ϊ ��
�� �ڢۢ����γɵļ����Ӱ뾶�ɴ�С��˳��Ϊ ��
�� ���⻯�ﳣ������Ԫ�آڵĹ������ﷴӦ�����ӷ���ʽΪ ��
�� �ٺ͢��γɵĵ���ɫ����ĵ���ʽΪ ��������ѧ��������Ϊ������Ӽ������ۼ����� ��
�� �ٺ͢ݵ�����������Ӧˮ�������Ӧ�����ӷ���ʽΪ ��
(16�֣�����ΪԪ�����ڱ��е�һ���֣���ע��������������ѧ�����ش��������⣺
|
��A |
��A |
��A |
��A |
��A |
��A |
��A |
0 |
|
|
2 |
|
|
|
|
|
�� |
�� |
|
|
3 |
�� |
�� |
�� |
|
|
|
|
�� |
|
4 |
�� |
�� |
|
|
|
|
|
|
�� 8��Ԫ���У���ѧ��������õ��� ���ǽ�������ǿ���� ��
�� �٢ڢݵ�����������Ӧˮ����ļ�����ǿ������˳��Ϊ ��
�� �ڢۢ����γɵļ����Ӱ뾶�ɴ�С��˳��Ϊ ��
�� ���⻯�ﳣ������Ԫ�آڵĹ������ﷴӦ�����ӷ���ʽΪ ��
�� �ٺ͢��γɵĵ���ɫ����ĵ���ʽΪ ��������ѧ��������Ϊ������Ӽ������ۼ����� ��
�� �ٺ͢ݵ�����������Ӧˮ�������Ӧ�����ӷ���ʽΪ ��