ĢāÄæÄŚČŻ
ŅŃÖŖ£ŗ CH3CH2CH2CH3(g)£«6.5O2(g) == 4CO2(g)£«5H2O(l) ¦¤H£½£2 878 kJ”¤mol-1
(CH3)2CHCH3(g)£«6.5O2(g) == 4CO2(g)£«5H2O(l) ¦¤H£½£2 869kJ”¤mol-1
ĻĀĮŠĖµ·ØÕżČ·µÄŹĒ£Ø £©
| A£®Õż¶”Ķé·Ö×Ó“¢“ęµÄÄÜĮæ“óÓŚŅģ¶”Ķé·Ö×Ó |
| B£®Õż¶”ĶéµÄĪČ¶ØŠŌ“óÓŚŅģ¶”Ķé |
| C£®Ņģ¶”Ķé×Ŗ»ÆĪŖÕż¶”ĶéµÄ¹ż³ĢŹĒŅ»øö·ÅČČ¹ż³Ģ |
| D£®Ņģ¶”Ķé·Ö×ÓÖŠµÄĢ¼Ēā¼ü±ČÕż¶”ĶéµÄ¶ą |
A
½āĪö

Į·Ļ°²įĻµĮŠ“š°ø
Ļą¹ŲĢāÄæ




 £¬WµÄČÜŅŗÄÜŹ¹pHŹŌÖ½±äŗģ£¬»¹ÄÜ·¢ÉśŅų¾µ·“Ó¦£®
£¬WµÄČÜŅŗÄÜŹ¹pHŹŌÖ½±äŗģ£¬»¹ÄÜ·¢ÉśŅų¾µ·“Ó¦£®

 ½į¹¹ĻąĖʵē¼²»Äܱ»Ńõ»Æ³ÉČ©»ņĖį£®”°ÖĒÄÜŠĶ”±“ó·Ö×Ó£¬ŌŚÉśĪļ¹¤³ĢÖŠÓŠ¹ć·ŗµÄÓ¦ÓĆĒ°¾°£®PMAA¾ĶŹĒŅ»ÖÖ”°ÖĒÄÜŠĶ”±“ó·Ö×Ó£¬æÉÓĆÓŚÉśĪļÖĘŅ©ÖŠ“ó·Ö×ÓÓėŠ”·Ö×ӵķ֥ė£®ĻĀĮŠŹĒŅŌĪļÖŹAĪŖĘšŹ¼·“Ó¦ĪļŗĻ³Éøß·Ö×Ó»ÆŗĻĪļPMAAµÄĀ·Ļߣŗ
½į¹¹ĻąĖʵē¼²»Äܱ»Ńõ»Æ³ÉČ©»ņĖį£®”°ÖĒÄÜŠĶ”±“ó·Ö×Ó£¬ŌŚÉśĪļ¹¤³ĢÖŠÓŠ¹ć·ŗµÄÓ¦ÓĆĒ°¾°£®PMAA¾ĶŹĒŅ»ÖÖ”°ÖĒÄÜŠĶ”±“ó·Ö×Ó£¬æÉÓĆÓŚÉśĪļÖĘŅ©ÖŠ“ó·Ö×ÓÓėŠ”·Ö×ӵķ֥ė£®ĻĀĮŠŹĒŅŌĪļÖŹAĪŖĘšŹ¼·“Ó¦ĪļŗĻ³Éøß·Ö×Ó»ÆŗĻĪļPMAAµÄĀ·Ļߣŗ



 +H2O
+H2O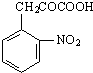 +C2H5OH
+C2H5OH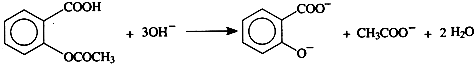
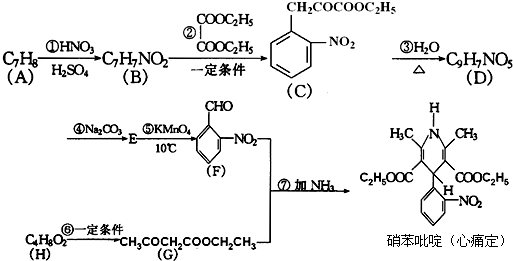
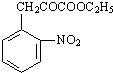
 ij·¼ĻćĢžXŹĒŅ»ÖÖÖŲŅŖµÄÓŠ»ś»Æ¹¤ŌĮĻ£¬ĖüµÄĻą¶Ō·Ö×ÓÖŹĮæĪŖ92£®ŃŠ¾æ²æĆÅŅŌXĪŖ³õŹ¼ŌĮĻÉč¼Ę³öČēĻĀ×Ŗ»Æ¹ŲĻµĶ¼£Ø²æ·Ö²śĪļ”¢ŗĻ³ÉĀ·Ļß”¢·“Ó¦Ģõ¼žĀŌČ„£©£¬ĘäÖŠAŹĒŅ»ĀČ“śĪļ£¬HŹĒŅ»ÖÖ¹¦ÄÜøß·Ö×Ó£¬Į“½Ś×é³ÉĪŖC7H5NO£®
ij·¼ĻćĢžXŹĒŅ»ÖÖÖŲŅŖµÄÓŠ»ś»Æ¹¤ŌĮĻ£¬ĖüµÄĻą¶Ō·Ö×ÓÖŹĮæĪŖ92£®ŃŠ¾æ²æĆÅŅŌXĪŖ³õŹ¼ŌĮĻÉč¼Ę³öČēĻĀ×Ŗ»Æ¹ŲĻµĶ¼£Ø²æ·Ö²śĪļ”¢ŗĻ³ÉĀ·Ļß”¢·“Ó¦Ģõ¼žĀŌČ„£©£¬ĘäÖŠAŹĒŅ»ĀČ“śĪļ£¬HŹĒŅ»ÖÖ¹¦ÄÜøß·Ö×Ó£¬Į“½Ś×é³ÉĪŖC7H5NO£®
 £Ø±½°·£¬Ņ×±»Ńõ»Æ£©
£Ø±½°·£¬Ņ×±»Ńõ»Æ£©


 ŗĶĘäĖūĪŽ»śĪļŗĻ³É
ŗĶĘäĖūĪŽ»śĪļŗĻ³É Éč¼Ę×īŗĻĄķµÄ·½°ø£¬Š“³öŗĻ³ÉĀ·Ļߣ؊č×¢Ć÷·“Ó¦Ģõ¼ž£©
Éč¼Ę×īŗĻĄķµÄ·½°ø£¬Š“³öŗĻ³ÉĀ·Ļߣ؊č×¢Ć÷·“Ó¦Ģõ¼ž£©
