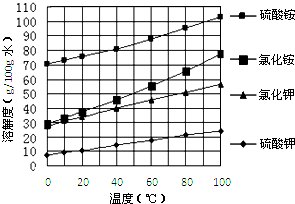
��Ŀ����
Ϊ��ģ�ҵ������������̣�ijС��ͬѧ���������װ�ã�������װ���ԣ���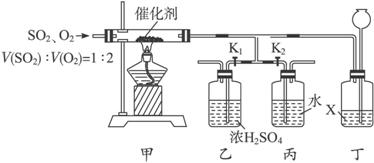
��1��д����������������Ӧ�Ļ�ѧ����ʽ_________________________________________��
��2���ڹ�ҵ�����У���װ�ü�������ͬ���豸������____________����װ����������ͬ���豸������____________��
��3����Ӧ��ͨ���V��SO2����V(O2)=1��2����Ŀ����___________________________��
��4��ʵ��ʱ�ȹر�K1����K2����һ����ٴ�K1���ر�K2����װ���Һͱ��в�����������____________��
��5����֪Һ��X�Ȳ����ܽ���������Ҳ�����ܽ���������װ�ö���������________________________��
��6������װ������һ�����Ե�ȱ�ݣ���ָ��____________________________________���Ľ��ķ�����____________________________________��
��1��4FeS2+11O2![]() 2Fe2O3+8SO2
2Fe2O3+8SO2
��2���Ӵ��� ������
��3��ͨ���ʵ�������O2�������SO2��������
��4����
��5��ʹװ���ڵ�ѹǿά����һ����Χ����ֹ�ҡ���װ���ڵ�Һ�嵹��������������Ҳ�ɣ�
��6��ȱ��β������װ�ã�Ӧ���ڱ�����������ʢ���ռ���Һ��ϴ��ƿ
����:��1������ѧ���Է�Ӧԭ�������ա�
��2������װ��ͼ��֪��������ʹSO2��O2�ڴ��������·���������Ӧ�����Լ���Ӵ���������ͬ������װ��Ũ����Ӧ��������������ͬ��
��3�����ݷ�Ӧԭ����֪�����������ɼ�Ŀ��������SO2��ת���ʡ�
��4��������Ϊˮ������SO3�����գ��ײ���������
��5��ʹװ���ڵ�ѹǿά����һ����Χ����ֹ�ҡ���װ���ڵ�Һ�嵹������ȫƿ�����á�
��6��ȱ��β������װ�ã�Ӧ���ڱ�����������ʢ ���ռ���Һ��ϴ��ƿ��������Ⱦ������

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д��Ի�����Ϊԭ��������������������к�Fe2O3��SiO2��Al2O3��MgO�ȡ�ʵ����ģ�ҵ���������Ʊ�����(Fe2O3)���������£�

��1���������ijɷ�������������������� �� д�����ܹ���Fe2O3��ϡ���ᷴӦ�����ӷ�Ӧ����ʽ�� ��
��2�����������У�Ϊ��ȷ������Ĵ��ȣ�����������Ҫ������Һ��pH�ķ�Χ��_________��������������������������ʽ����ʱ��Һ��pH���±���
|
������ |
Fe(OH)3 |
Al(OH)3 |
Fe(OH)2 |
Mg(OH)2 |
|
��ʼ���� |
2.7 |
3.8 |
7.5 |
9.4 |
|
��ȫ���� |
3.2 |
5.2 |
9.7 |
12.4 |
��3������A����Ҫ�ɷ�Ϊ ����ҺB���Ի��յ�������______________________��
��4������ϴ�ӹ��̵�ʵ����� ��
��5����֪����������Ϊw kg�����������Ʊ������У���Ԫ�����25%�����յõ����������Ϊm kg����ԭ������������Ԫ����������Ϊ ��������������ʽ��ʾ����
����֪���ԭ��������O 16 S 32 Fe 56 ��
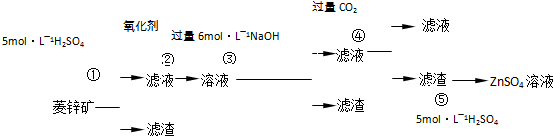
 ��2009?����ģ�⣩����ݹ�ҵ��������й�֪ʶ�ش��������⣺
��2009?����ģ�⣩����ݹ�ҵ��������й�֪ʶ�ش��������⣺