ĚâÄżÄÚČÝ
ŇŃÖŞŁş CH3CH2CH2CH3(g)Ł«6.5O2(g)== 4CO2(g)Ł«5H2O(l)Ł»DH =Ł2878 kJˇ¤molŁ1
(CH3)2CHCH3(g)Ł«6.5O2(g)== 4CO2(g)Ł«5H2O(l)Ł»DH =Ł2869 kJˇ¤molŁ1
ĎÂÁĐ˵·¨ŐýČ·µÄĘÇŁş
AŁ®Őý¶ˇÍé·Ö×Ó´˘´ćµÄÄÜÁż´óÓÚŇ춡Íé·Ö×Ó
BŁ®Őý¶ˇÍéµÄÎȶ¨ĐÔ´óÓÚŇ춡Íé
CŁ®Ň춡Íéת»ŻÎŞŐý¶ˇÍéµÄąýłĚĘÇŇ»¸ö·ĹČČąýłĚ
DŁ®Ň춡Íé·Ö×ÓÖеÄĚĽÇâĽü±ČŐý¶ˇÍéµÄ¶ŕ
A

Á·Ď°˛áϵÁĐ´đ°¸
 żě˝ÝÓ˘ÓďÖÜÖÜÁ·ĎµÁĐ´đ°¸
żě˝ÝÓ˘ÓďÖÜÖÜÁ·ĎµÁĐ´đ°¸
ĎŕąŘĚâÄż




 Ł¬WµÄČÜŇşÄÜĘąpHĘÔÖ˝±äşěŁ¬»ąÄÜ·˘ÉúŇřľµ·´Ó¦Ł®
Ł¬WµÄČÜŇşÄÜĘąpHĘÔÖ˝±äşěŁ¬»ąÄÜ·˘ÉúŇřľµ·´Ó¦Ł®

 ˝áąąĎŕËƵĴĽ˛»Äܱ»Ńő»ŻłÉČ©»ňËᣮˇ°ÖÇÄÜĐ͡±´ó·Ö×ÓŁ¬ÔÚÉúÎ﹤łĚÖĐÓĐąă·şµÄÓ¦ÓĂÇ°ľ°Ł®PMAAľÍĘÇŇ»ÖÖˇ°ÖÇÄÜĐ͡±´ó·Ö×ÓŁ¬żÉÓĂÓÚÉúÎďÖĆŇ©ÖĐ´ó·Ö×ÓÓëС·Ö×ӵķÖŔ룮ĎÂÁĐĘÇŇÔÎďÖĘAÎŞĆđĘĽ·´Ó¦ÎďşĎłÉ¸ß·Ö×Ó»ŻşĎÎďPMAAµÄ·Ďߣş
˝áąąĎŕËƵĴĽ˛»Äܱ»Ńő»ŻłÉČ©»ňËᣮˇ°ÖÇÄÜĐ͡±´ó·Ö×ÓŁ¬ÔÚÉúÎ﹤łĚÖĐÓĐąă·şµÄÓ¦ÓĂÇ°ľ°Ł®PMAAľÍĘÇŇ»ÖÖˇ°ÖÇÄÜĐ͡±´ó·Ö×ÓŁ¬żÉÓĂÓÚÉúÎďÖĆŇ©ÖĐ´ó·Ö×ÓÓëС·Ö×ӵķÖŔ룮ĎÂÁĐĘÇŇÔÎďÖĘAÎŞĆđĘĽ·´Ó¦ÎďşĎłÉ¸ß·Ö×Ó»ŻşĎÎďPMAAµÄ·Ďߣş



 +H2O
+H2O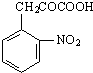 +C2H5OH
+C2H5OH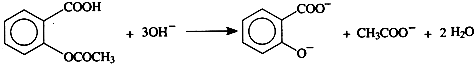
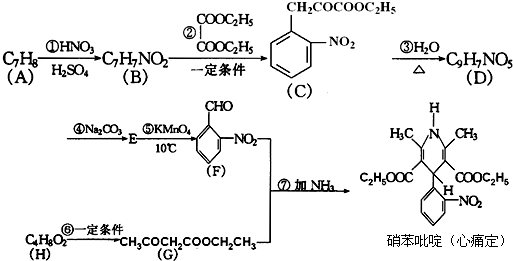
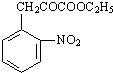
 Äł·ĽĎăĚţXĘÇŇ»ÖÖÖŘŇŞµÄÓĐ»ú»Żą¤ÔÁĎŁ¬ËüµÄĎŕ¶Ô·Ö×ÓÖĘÁżÎŞ92Ł®ŃĐľż˛żĂĹŇÔXÎŞłőĘĽÔÁĎÉčĽĆłöČçĎÂת»ŻąŘϵͼŁ¨˛ż·Ö˛úÎşĎłÉ·Ďߡ˘·´Ó¦ĚőĽţÂÔČĄŁ©Ł¬ĆäÖĐAĘÇŇ»ÂČ´úÎHĘÇŇ»ÖÖą¦Äܸ߷Ö×ÓŁ¬Á´˝Ú×éłÉÎŞC7H5NOŁ®
Äł·ĽĎăĚţXĘÇŇ»ÖÖÖŘŇŞµÄÓĐ»ú»Żą¤ÔÁĎŁ¬ËüµÄĎŕ¶Ô·Ö×ÓÖĘÁżÎŞ92Ł®ŃĐľż˛żĂĹŇÔXÎŞłőĘĽÔÁĎÉčĽĆłöČçĎÂת»ŻąŘϵͼŁ¨˛ż·Ö˛úÎşĎłÉ·Ďߡ˘·´Ó¦ĚőĽţÂÔČĄŁ©Ł¬ĆäÖĐAĘÇŇ»ÂČ´úÎHĘÇŇ»ÖÖą¦Äܸ߷Ö×ÓŁ¬Á´˝Ú×éłÉÎŞC7H5NOŁ®
 Ł¨±˝°·Ł¬Ň×±»Ńő»ŻŁ©
Ł¨±˝°·Ł¬Ň×±»Ńő»ŻŁ©


 şÍĆäËűÎŢ»úÎďşĎłÉ
şÍĆäËűÎŢ»úÎďşĎłÉ ÉčĽĆ×îşĎŔíµÄ·˝°¸Ł¬Đ´łöşĎłÉ·Ďߣ¨ĐčעĂ÷·´Ó¦ĚőĽţŁ©
ÉčĽĆ×îşĎŔíµÄ·˝°¸Ł¬Đ´łöşĎłÉ·Ďߣ¨ĐčעĂ÷·´Ó¦ĚőĽţŁ©
