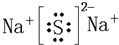��Ŀ����
�������ж�����Ԫ�����ʵ����ݣ�Ԫ�ر�� Ԫ������ | �� | �� | �� | �� | �� | �� | �� | �� |
ԭ�Ӱ뾶(10-10 m) | 0.74 | 1.60 | 1.52 | 1.10 | 0.99 | 1.86 | 0.75 | 0.82 |
�����ͻ��ϼ� |
| +2 | +1 | +5 | +7 | +1 | +5 | +3 |
-2 |
|
| -3 | -1 |
| -3 |
|
�Իش��������⣺
(1)����Ԫ���д���ͬһ�������____________��Ԫ�آ������ڱ��е�λ��Ϊ____________��
(2)�ϱ���ij����Ԫ���γɵķ����У�ÿ��ԭ�Ӷ����������Ϊ8���ӵ��ȶ��ṹ��д�������ʽ____________��
(3)Ԫ�آ٢����γ����ֻ����д�����н��ȶ��Ļ�������ˮ��Ӧ�����ӷ���ʽ��__________________________________________________________________��
����������Ԫ�����ʵ����ݣ�����Ԫ�������ɵĵݱ���������ã���ΪO����ΪMg����ΪLi����ΪP����ΪCl����ΪNa����ΪN����ΪB��
(1)��ԭ�Ӱ뾶����Ҫ���ϼ�����֪��ͬһ����Ԫ��Ϊ�ۺ͢ޣ��ܺ͢ߣ��ں�Ԫ��λ�ڵ������ڵڢ�A�壻
(2)���������Ϊ8���ӵ��ȶ��ṹ�У�
(3)�٢����γ�Na2O��Na2O2��Na2O2���ȶ�����ˮ���������ӷ���ʽ��2Na2O2+2H2O![]() 4Na++4OH-+O2��
4Na++4OH-+O2��
�𰸣�(1)����ޡ������ ��3���ڵڢ�A��
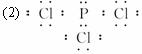
(3)2Na2O2+2H2O![]() 4Na++4OH-+O2��
4Na++4OH-+O2��

��ϰ��ϵ�д�
�����Ŀ