��Ŀ����
Ϊ��ȥ�����е�Ca2+��Mg2+��SO42-�Լ���ɳ�����ʣ�ijͬѧ�����һ���Ʊ����ε�ʵ�鷽�����������£����ڳ������Լ��Թ�����
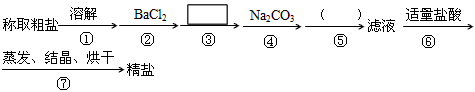
��1���ж�BaCl2�ѹ����ķ�����_____________________��
��2���ڢܲ��У�д����Ӧ�Ļ�ѧ����ʽ���������Һ��Ca2+����Ҫ������ʽΪCaCl2�� ��
___________________________________��
��3���� ����д��ʹ�ó����Լ��Ļ�ѧʽ__________���ڣ� ���еIJ���������_______��
����д��ʹ�ó����Լ��Ļ�ѧʽ__________���ڣ� ���еIJ���������_______��
��4����ʵ����Ʒ����Ż��ĽǶȷ�������ںܿ͢ɷ�ߵ�____________����ǡ������������˵�����ɡ���ԭ���ǣ�_____________________������ۺܿ͢ɷ�ߵ�____________��
��5�������������ٽ��в����ݣ�����ʵ��������Ӱ�죬��ԭ���ǣ�____________________________
��2���ڢܲ��У�д����Ӧ�Ļ�ѧ����ʽ���������Һ��Ca2+����Ҫ������ʽΪCaCl2�� ��
___________________________________��
��3����
 ����д��ʹ�ó����Լ��Ļ�ѧʽ__________���ڣ� ���еIJ���������_______��
����д��ʹ�ó����Լ��Ļ�ѧʽ__________���ڣ� ���еIJ���������_______����4����ʵ����Ʒ����Ż��ĽǶȷ�������ںܿ͢ɷ�ߵ�____________����ǡ������������˵�����ɡ���ԭ���ǣ�_____________________������ۺܿ͢ɷ�ߵ�____________��
��5�������������ٽ��в����ݣ�����ʵ��������Ӱ�죬��ԭ���ǣ�____________________________
��1��ȡ�ڢڲ�����ϲ���Һ1��2���ڵ�ΰ��ϣ���ȡ�����ϲ���Һ���Թ��У����ٵ���1��2��BaCl2��Һ������Һδ����ǣ������BaCl2�ѹ����������𰸽Կɣ�
��2��CaCl2+Na2CO3==CaCO3��+2NaCl��BaCl2+Na2CO3==BaCO3��+2NaCl
��3��NaOH������
��4��������BaCl2����Ҫ��Na2CO3��ȥ�� �ǣ�
��5�����в��ֳ����ܽ⣬�Ӷ�Ӱ���Ƶþ��εĴ���
��2��CaCl2+Na2CO3==CaCO3��+2NaCl��BaCl2+Na2CO3==BaCO3��+2NaCl
��3��NaOH������
��4��������BaCl2����Ҫ��Na2CO3��ȥ�� �ǣ�
��5�����в��ֳ����ܽ⣬�Ӷ�Ӱ���Ƶþ��εĴ���

��ϰ��ϵ�д�
 ���Ӣ��������ϵ�д�
���Ӣ��������ϵ�д�
�����Ŀ


 Ϊ��ȥ�����е�Ca2+��Mg2+��Fe3+��SO42-�Լ���ɳ�����ʣ�ijͬѧ�����һ���Ʊ����ε�ʵ�鷽�����������£����ڳ������Լ��Թ�������
Ϊ��ȥ�����е�Ca2+��Mg2+��Fe3+��SO42-�Լ���ɳ�����ʣ�ijͬѧ�����һ���Ʊ����ε�ʵ�鷽�����������£����ڳ������Լ��Թ�������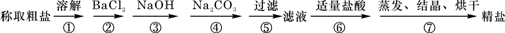

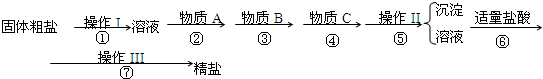
 aOH��Һ b��KOH��Һ c��BaCl2��Һ d��Ba��NO3��2��Һ
aOH��Һ b��KOH��Һ c��BaCl2��Һ d��Ba��NO3��2��Һ