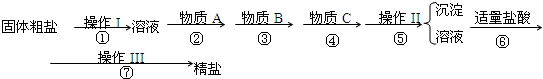
��Ŀ����
Ϊ��ȥ�����е�Ca2+��Mg2+��SO42-�Լ���ɳ�����ʣ�ijͬѧ�����һ���Ʊ����ε�ʵ�鷽�����������£����ڳ������Լ��Թ�������
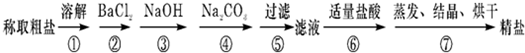
��1���ж�BaCl2�ѹ����ķ�����
��2���ڢڲ��У��йص����ӷ���ʽ��
��3�����ȼ����������ٹ��ˣ�����ʵ��������Ӱ�죬��ԭ���ǣ�
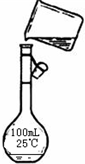
��4��Ϊ���龫�δ��ȣ�������1000mL.0.2mol/L NaCl�����Σ���Һ��������ʱ�۲�Һ�����ӣ�����������Ƶ���ҺŨ�ȣ��ƫ�ߡ���ƫ�͡���

��1���ж�BaCl2�ѹ����ķ�����
ȡ���������ϲ���Һ���Թ��У������еμ�BaCl2��Һ����������������BaCl2��Һ�ѹ���
ȡ���������ϲ���Һ���Թ��У������еμ�BaCl2��Һ����������������BaCl2��Һ�ѹ���
����2���ڢڲ��У��йص����ӷ���ʽ��
Ba2++SO42-=BaSO4��
Ba2++SO42-=BaSO4��
���ڢ۲��У��йص����ӷ���ʽ��Mg2++2OH-=Mg��OH��2��
Mg2++2OH-=Mg��OH��2��
����3�����ȼ����������ٹ��ˣ�����ʵ��������Ӱ�죬��ԭ���ǣ�
���в��ֳ�����CaCO3��BaCO3�ܽ⣬�Ӷ�Ӱ���Ƶþ��εĴ���
���в��ֳ�����CaCO3��BaCO3�ܽ⣬�Ӷ�Ӱ���Ƶþ��εĴ���
����4��Ϊ���龫�δ��ȣ�������1000mL.0.2mol/L NaCl�����Σ���Һ��������ʱ�۲�Һ�����ӣ�����������Ƶ���ҺŨ�ȣ��ƫ�ߡ���ƫ�͡���
ƫС
ƫС
����������1�������Ȼ�����Һ��������������Ƿ���������ж��Ȼ����Ƿ������
��2���ڲ��Ǽ����Ȼ���������������ӣ���Ӧ�����ӷ���ʽΪ��Ba2++SO42-=BaSO4�����۲��Ǽ����������Ƴ���þ���ӣ���Ӧ���ӷ���ʽΪ��Mg2++2OH-=Mg��OH��2����
��3�����ȼ����������ٹ��ˣ����ɵ�̼��ơ�̼�ᱵ�������ܽ�������ﲻ���ᴿ�Ȼ��Ƶ����ã�
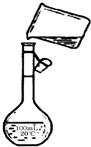
��4����������ƿ���Կ̶ȶ�Һ���ԭ��������
��2���ڲ��Ǽ����Ȼ���������������ӣ���Ӧ�����ӷ���ʽΪ��Ba2++SO42-=BaSO4�����۲��Ǽ����������Ƴ���þ���ӣ���Ӧ���ӷ���ʽΪ��Mg2++2OH-=Mg��OH��2����
��3�����ȼ����������ٹ��ˣ����ɵ�̼��ơ�̼�ᱵ�������ܽ�������ﲻ���ᴿ�Ȼ��Ƶ����ã�
��4����������ƿ���Կ̶ȶ�Һ���ԭ��������
����⣺��1����������Ȼ�����Һ��ȥ��������ӣ���������������ѳ������ɾ�ֹƬ�����ϲ���Һ�����μ�һ���Ȼ�����Һ�������ֻ��Ǿ�˵������������Ѿ�������BaCl2��Һ�ѹ�����
�ʴ�Ϊ��ȡ���������ϲ���Һ���Թ��У������еμ�BaCl2��Һ����������������BaCl2��Һ�ѹ��������������𰸾��ɣ���
��2���ڲ��Ǽ����Ȼ���������������ӣ���Ӧ�����ӷ���ʽΪ��Ba2++SO42-=BaSO4�����۲��Ǽ����������Ƴ���þ���ӣ���Ӧ���ӷ���ʽΪ��Mg2++2OH-=Mg��OH��2����
�ʴ�Ϊ��Ba2++SO42-=BaSO4����Mg2++2OH-=Mg��OH��2����
��3�����ȼ����������ٹ��ˣ����ɵij�����CaCO3��BaCO3���ܽ��������У�Ӱ���Ƶþ��εĴ��ȣ��ʴ𰸣����в��ֳ�����CaCO3��BaCO3�ܽ⣬�Ӷ�Ӱ���Ƶþ��εĴ��ȣ�
��4������ƿ������Һ����ʱ�����������ݿ̶ȶ�Һ������жϣ����۲�Һ�����ӣ������ˮ���ᳬ���̶�����������Ƶ���ҺŨ��ƫС���ʴ�Ϊ��ƫС��
�ʴ�Ϊ��ȡ���������ϲ���Һ���Թ��У������еμ�BaCl2��Һ����������������BaCl2��Һ�ѹ��������������𰸾��ɣ���
��2���ڲ��Ǽ����Ȼ���������������ӣ���Ӧ�����ӷ���ʽΪ��Ba2++SO42-=BaSO4�����۲��Ǽ����������Ƴ���þ���ӣ���Ӧ���ӷ���ʽΪ��Mg2++2OH-=Mg��OH��2����
�ʴ�Ϊ��Ba2++SO42-=BaSO4����Mg2++2OH-=Mg��OH��2����
��3�����ȼ����������ٹ��ˣ����ɵij�����CaCO3��BaCO3���ܽ��������У�Ӱ���Ƶþ��εĴ��ȣ��ʴ𰸣����в��ֳ�����CaCO3��BaCO3�ܽ⣬�Ӷ�Ӱ���Ƶþ��εĴ��ȣ�
��4������ƿ������Һ����ʱ�����������ݿ̶ȶ�Һ������жϣ����۲�Һ�����ӣ������ˮ���ᳬ���̶�����������Ƶ���ҺŨ��ƫС���ʴ�Ϊ��ƫС��
���������⿼���˴����ᴿ��ʵ�鷽����ע�����⡢�������ʵ�Ӧ�ã��ؼ��dz����Լ��ļ���˳���жϣ���Ҫ�������Լ�������ڳ�ȥ������ƿ��ʹ�÷��������������ۺ��Խ�ǿ����Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
 ���100��1�ž�ϵ�д�
���100��1�ž�ϵ�д�
�����Ŀ
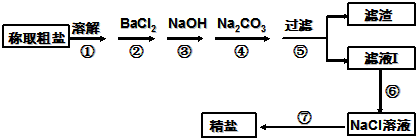
 Ϊ��ȥ�����е�Ca2+��Mg2+��Fe3+��SO42-�Լ���ɳ�����ʣ�ijͬѧ�����һ���Ʊ����ε�ʵ�鷽�����������£����ڳ������Լ��Թ�������
Ϊ��ȥ�����е�Ca2+��Mg2+��Fe3+��SO42-�Լ���ɳ�����ʣ�ijͬѧ�����һ���Ʊ����ε�ʵ�鷽�����������£����ڳ������Լ��Թ�������
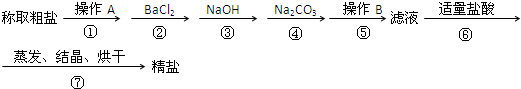
 aOH��Һ b��KOH��Һ c��BaCl2��Һ d��Ba��NO3��2��Һ
aOH��Һ b��KOH��Һ c��BaCl2��Һ d��Ba��NO3��2��Һ