��Ŀ����
��(Sn)��һ����Ҫ�Ľ������������������������ϡ��뵼�塣��Ԫ��λ�����ڱ���5���ڣ���IVA�塣
�����ڻ������г����Ļ��ϼ������֣�+2��_________��
��������������Ҳ����NaOH��Һ��Ӧ�����ɵ��ζ��Ǻ�+2�۵�����д������NaOH��Һ��Ӧ�����ӷ���ʽ_____________________________��
����ͼ�ǹ�ҵұ�����Ļ������̣�
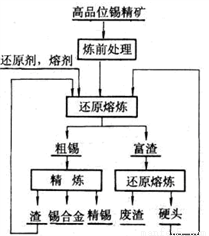
����ǰ����������������Ҫ����������ͭ��Ǧ��������黯��ڿ����б��ռ����̣�ʹ�����Ԫ��ת��Ϊ�ӷ���SO2��As2O3����д����Ż�����FeAsS2���������ɺ���ɫ����Ļ�ѧ����ʽ______________�����ɵ������ù�����NaOH��Һ���գ�����Һ�е�Ũ�����ĺ������������_______
�ڻ�ԭ�����ķ�Ӧԭ��Ϊ��SnO2(s)+2CO(g) Sn(s)+2CO2(g)��һ���¶��£��ڹ̶��ݻ����ܱ������У�SnO2��CO��Ӧ��ƽ��������������ƽ��Ħ������Ϊ37.6�����¶��µ�ƽ�ⳣ��K=____________
Sn(s)+2CO2(g)��һ���¶��£��ڹ̶��ݻ����ܱ������У�SnO2��CO��Ӧ��ƽ��������������ƽ��Ħ������Ϊ37.6�����¶��µ�ƽ�ⳣ��K=____________
�۴�������Ҫ����Fe��Cu��Pb��Sb�����������õ�⾫��������þ���������SnSO4��Һ�����Һ���������ӵ�Դ��______��������֪�����������к���Sb��Cu��PbSO4�ȣ���������������缫��ӦʽΪ________________________________________��
�ܾ����������������ԭ����������Ӳͷ��һ�����е�Ԫ����________��