��Ŀ����
��9�֣� ��֪ij��Һ��ֻ����OH����H+��NH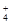 ��Cl���������ӣ�ijͬѧ�Ʋ�������Ũ�ȴ�С˳�����������ֹ�ϵ��
��Cl���������ӣ�ijͬѧ�Ʋ�������Ũ�ȴ�С˳�����������ֹ�ϵ��
��c��Cl������c��NH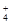 ����c��H+����c��OH����
����c��H+����c��OH����
��c��Cl������c��NH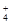 ����c��OH������c��H+��
����c��OH������c��H+��
��c��NH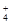 ����c��Cl������c��OH������c��H+��
����c��Cl������c��OH������c��H+��
��c��Cl������c��H+����c��NH ����c��OH����
����c��OH����
��д���пհף�
��1������Һ��ֻ�ܽ���һ�����ʣ����������_________��������������Ũ�ȵĴ�С˳��Ϊ________������ţ���
��2����������ϵ�Т�����ȷ�ģ�����Һ�е�����Ϊ_____________����������ϵ�Т�����ȷ�ģ�����Һ�е�����Ϊ____ ___��
(3)������ϵ���Ƿ���ȷ:____________(����ȷ����ȷ)��_________________(��ȷ��ָ�����ʣ�����ȷ��˵��ԭ��)
��4��������Һ���������ȵ�ϡ����Ͱ�ˮ��϶��ɣ���ǡ�ó����ԣ�����ǰc��HCl��________c��NH3��H2O��������ڡ���С�ڡ����ڡ�����ͬ�������ǰ����c��H+���ͼ���c��OH�����Ĺ�ϵ��c��H+��_______c��OH������
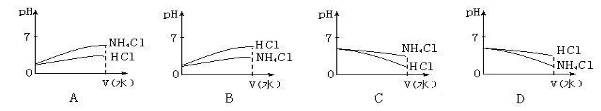
��5����pH��ͬ��NH4Cl��Һ��HCl��Һϡ����ͬ�ı�����������ͼ����ȷ���ǣ���ͼ����ţ�________________��
��1��NH4Cl �� ��2�� NH4Cl��NH3.H2O NH4Cl��HCl ��3���� Υ������غ�
(4) �� ���� (5)B
����������

