��Ŀ����
��13�֣�X��Y��WΪԭ���������ε����Ķ�����Ԫ�أ�X��Yͬ���壬Y��W����̬�⻯�������ͬ�ĵ�������X�ĵ���ֻ�������ԡ�
��1��д��ʵ������ȡW2��Ӧ�����ӷ���ʽ__________________________��
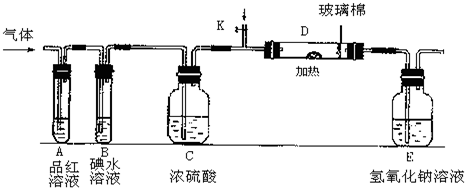
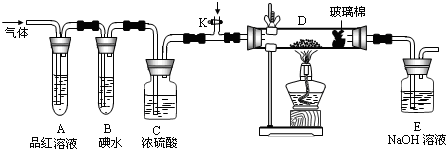
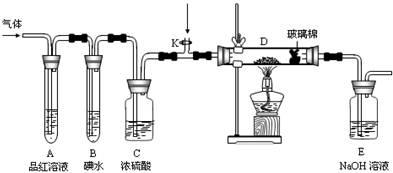
��2��ijС�������ͼ��ʾ��װ��ͼ��ͼ�мгֺͼ���װ����ȥ�����ֱ��о�YX2��W2�����ʡ�
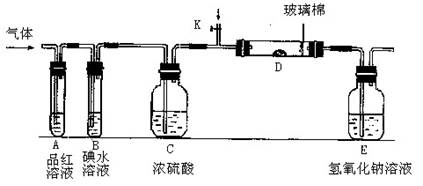
�ٷֱ�ͨ��YX2��W2����װ��A�й۲쵽�������Ƿ���ͬ�����ͬ������ͬ����__________����װ��D��װ�������ۣ���ͨ��W2ʱ�۲쵽������Ϊ_______________________����װ��D��װ���Ǵ�������ͨ��YX2ʱ����Kͨ������X2����ѧ��Ӧ����ʽΪ__________________________________��
����װ��B��װ��5.0mL 1.0��10-3mol/L�ĵ�ˮ����ͨ������W2��ȫ��Ӧ��ת����5.0��10-5mol���ӣ��÷�Ӧ�Ļ�ѧ����ʽΪ__________________��
��3��ijͬѧ��������YX2ͨ��һ֧װ���Ȼ�����Һ���Թܣ�δ���������ɣ�����Թ��м������������ĸ��________________�����Կ�����ɫ�������ɣ���ԭ��ֱ�Ϊ_________________________________________________��
A����ˮ B��ϡ���� C��ϡ���� D���Ȼ���
��4������Ԫ��Y��X��ɡ�2�����Z��Z��Y��X��������ΪY��X=4��3����W2�뺬Z����Һ��ȫ��Ӧ����dz��ɫ����������ȡ�ϲ���Һ�����Ȼ�����Һ���а�ɫ�����������÷�Ӧ�����ӷ���ʽΪ____________________________��
��1��MnO2+4H
-+2Cl- Mn2++Cl2��+2H2O��������������������2��
Mn2++Cl2��+2H2O��������������������2��
��2������ͬ �����ػ�ɫ���̡���������������������������������2��
2SO2+O2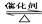 2SO2��������������������������������������2��
2SO2��������������������������������������2��
��5Cl2+l2+6H2O=2HlO3+10HCl������������������������2��
��3��A C �����백ˮʱ��ʹ����Һ��Ϊ���ԣ�����BaSO3��ɫ������������HNO3ʱ��ʹ��Һ��H2SO3������H2SO4������BaSO4��ɫ����������������3��
��4��S2O23-+Cl2+H2O=SO2-4+S��2Cl-+2H-��������������������2��
��������

 ��ڽ��ȫ������ϵ�д�
��ڽ��ȫ������ϵ�д�