��Ŀ����
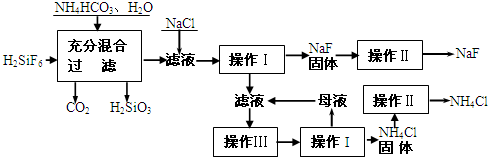
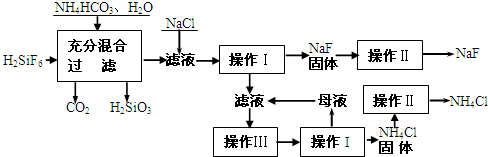
��������һ������ũ����ɱ����ɱ�����ľ�ĵķ�������ʵ���ҿ�ͨ����ͼ��ʾ�������Է����ᣨH2SiF6��������Ϊԭ����ȡ�����ƣ����õ�����Ʒ�Ȼ�泥�

��֪��20��ʱ�Ȼ�淋��ܽ��Ϊ37��2g�������Ƶ��ܽ��Ϊ2g��Na2SiF6����ˮ��
��1�����̢���H2SiF6��NH4HCO3��Ӧ�õ���NH4F��H2SiO3��ɻ���Һ����д�����̢�����ط�Ӧ�Ļ�ѧ����ʽ���� ���ܹ�����������Ӧ��ԭ�� ��
��2������I��IV����ͬ�ģ���ָ�������� ��
��3������II��ϴ�ӳ����Ĺ����ǽ�©����ĸҺȫ���˳���
;
��4�����̢���NH4HCO3�����������ԭ���� ��
��1�� �� NH4F+NaCl==NaF��+NH4Cl��NaF ���ܽ�ȱ�NH4F���ܽ��С�ö�
��2������
��3��������ֽ�ϼ�����ˮ��ǡ�ý�û���壬���������˳����ظ�����2-3�Σ��Գ�ȥ������������
��4�����뱣֤����H2SiF6��ȫ����Ӧ����ֹ���¢�������Na2SiF6����

 ��ͼͼ�麮����ҵ������ҵ���ִ�ѧ������ϵ�д�
��ͼͼ�麮����ҵ������ҵ���ִ�ѧ������ϵ�д�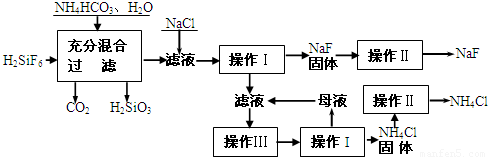
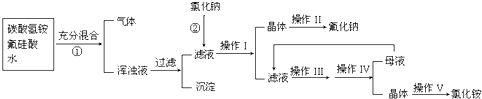
�й�������ˮ���ܽ�ȼ��±�����ش��������⣺
| �¶� | 10�� | 20�� | 30�� | �ܽ�ȣ�20��NaF-4g��0��NH4F-100g�� ����Na2SiF6-����ˮ |
| NH4Cl�ܽ�� | 33.3g | 37.2g | 41.4g |
��2�����������з���������ѧ��Ӧ����һ����Ӧ�Ļ�ѧ����ʽΪ��______���ڶ�����Ӧ�Ļ�ѧ����ʽΪ______��
��3������II��������______��������ľ��������______��
��4��������NH4HCO3�����������ԭ����______��
��5��NaF�����������������࣬ʹ�ú����������Ԥ��ȣ�ݣ���˵������NaF��������ν��ǻ������[Ca5��PO4��3��OH��]ת��Ϊ�����ܵķ������[Ca5��PO4��3F]______��