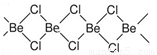
��Ŀ����
��������Ϊ ������ӵ���ѱ��㷺������Яʽ��Դ�����ܵ���Դ�ѷ����������һ����չ��
������ӵ���ѱ��㷺������Яʽ��Դ�����ܵ���Դ�ѷ����������һ����չ��
��1�����ʯ�� ��һ��DZ�ڵ�����ӵ���������ϣ�������ͨ��
��һ��DZ�ڵ�����ӵ���������ϣ�������ͨ�� ��
�� ��
��
 ��Һ������������Ӧ�����ó�����80����ո�����³��Ͷ��Ƶá�
��Һ������������Ӧ�����ó�����80����ո�����³��Ͷ��Ƶá�
�ٹ�������ӦͶ��ʱ������ ��
�� ��Һֱ�ӻ�ϵ�ԭ����____________________��
��Һֱ�ӻ�ϵ�ԭ����____________________��
�ڹ�������Ӧ�Ļ�ѧ����ʽΪ__________________________��
�۸��³���ǰ������ �м�����������̿�ڣ������ó��˿��Ը��Ƴ��ͺ��
�м�����������̿�ڣ������ó��˿��Ը��Ƴ��ͺ�� �ĵ��������⣬����___________________________��
�ĵ��������⣬����___________________________��
��2���Ͼ�����ӵ�ص�����������������Ҫ���� ������Al��Fe�ȣ���ͨ������ʵ�鷽�������ܡ�ﮡ�
������Al��Fe�ȣ���ͨ������ʵ�鷽�������ܡ�ﮡ�
 ������ӵ���ѱ��㷺������Яʽ��Դ�����ܵ���Դ�ѷ����������һ����չ��
������ӵ���ѱ��㷺������Яʽ��Դ�����ܵ���Դ�ѷ����������һ����չ����1�����ʯ��
 ��һ��DZ�ڵ�����ӵ���������ϣ�������ͨ��
��һ��DZ�ڵ�����ӵ���������ϣ�������ͨ�� ��
�� ��
�� ��Һ������������Ӧ�����ó�����80����ո�����³��Ͷ��Ƶá�
��Һ������������Ӧ�����ó�����80����ո�����³��Ͷ��Ƶá� �ٹ�������ӦͶ��ʱ������
 ��
�� ��Һֱ�ӻ�ϵ�ԭ����____________________��
��Һֱ�ӻ�ϵ�ԭ����____________________�� �ڹ�������Ӧ�Ļ�ѧ����ʽΪ__________________________��
�۸��³���ǰ������
 �м�����������̿�ڣ������ó��˿��Ը��Ƴ��ͺ��
�м�����������̿�ڣ������ó��˿��Ը��Ƴ��ͺ�� �ĵ��������⣬����___________________________��
�ĵ��������⣬����___________________________����2���Ͼ�����ӵ�ص�����������������Ҫ����
 ������Al��Fe�ȣ���ͨ������ʵ�鷽�������ܡ�ﮡ�
������Al��Fe�ȣ���ͨ������ʵ�鷽�������ܡ�ﮡ� 
�� �������ܽ�����У� ��������
�������� ��
�� ���ܽ�����з�Ӧ�Ļ�ѧ����ʽΪ______________________________��
���ܽ�����з�Ӧ�Ļ�ѧ����ʽΪ______________________________��
�� �ڿ����м���ʱ��������������¶ȵı仯����ͼ��ʾ����֪�ܵ��������������290��ʱ����ȫ��ˮ����1000��ʱ��ʣ�����ijɷ�Ϊ_____________________�����ѧʽ������350~400�淶Χ�ڣ�ʣ�����ijɷ�Ϊ_________________�����ѧʽ��
�ڿ����м���ʱ��������������¶ȵı仯����ͼ��ʾ����֪�ܵ��������������290��ʱ����ȫ��ˮ����1000��ʱ��ʣ�����ijɷ�Ϊ_____________________�����ѧʽ������350~400�淶Χ�ڣ�ʣ�����ijɷ�Ϊ_________________�����ѧʽ��
 ��������
�������� ��
�� ���ܽ�����з�Ӧ�Ļ�ѧ����ʽΪ______________________________��
���ܽ�����з�Ӧ�Ļ�ѧ����ʽΪ______________________________�� ��
 �ڿ����м���ʱ��������������¶ȵı仯����ͼ��ʾ����֪�ܵ��������������290��ʱ����ȫ��ˮ����1000��ʱ��ʣ�����ijɷ�Ϊ_____________________�����ѧʽ������350~400�淶Χ�ڣ�ʣ�����ijɷ�Ϊ_________________�����ѧʽ��
�ڿ����м���ʱ��������������¶ȵı仯����ͼ��ʾ����֪�ܵ��������������290��ʱ����ȫ��ˮ����1000��ʱ��ʣ�����ijɷ�Ϊ_____________________�����ѧʽ������350~400�淶Χ�ڣ�ʣ�����ijɷ�Ϊ_________________�����ѧʽ��
��1����Fe2+�ڼ��������¸��ױ��������������𰸾��ɣ�����(NH4)2Fe(SO4)2+LiOH+H3PO4==LiFePO4
+2NH4HSO4+H2O�����������O2��Ӧ����ֹLiFePO4�е�Fe2+������
��2����8LiCoO2+Na2S2O3+11H2SO4==4LiSO4+8CoSO4+Na2SO4+11H2O
��CoO��Co2O3��Co3O4
+2NH4HSO4+H2O�����������O2��Ӧ����ֹLiFePO4�е�Fe2+������
��2����8LiCoO2+Na2S2O3+11H2SO4==4LiSO4+8CoSO4+Na2SO4+11H2O
��CoO��Co2O3��Co3O4

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
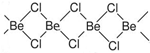 ﮺��뵥�ʼ�����������ʾ��������ԣ�
﮺��뵥�ʼ�����������ʾ��������ԣ�