��Ŀ����
��֪�����Ȼ�ѧ����ʽ��
��CH3COOH(l)��2O2(g)��2CO2(g)��2H2O(l)����H1����870.3 kJ/mol
��C(s)��O2(g)��CO2(g)����H2����393.5 kJ/mol
��H2(g)��1/2O2(g)��H2O(l)����H3����285.8 kJ/mol
д����C(s)��H2(g)��O2(g)��������CH3COOH(l)���Ȼ�ѧ����ʽ________��
�𰸣�
������
������
|
2C(s)��2H2(g)��O2(g)��CH2COOH(l)����H����488.3 kJ/mol |

��ϰ��ϵ�д�
 ������������Ӧ����ϵ�д�
������������Ӧ����ϵ�д� ͬ����չ�Ķ�ϵ�д�
ͬ����չ�Ķ�ϵ�д�
�����Ŀ
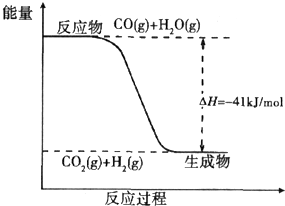 ú̿����ת��Ϊ�����Դ�ͻ���ԭ�ϣ�
ú̿����ת��Ϊ�����Դ�ͻ���ԭ�ϣ�