��Ŀ����
���Ǵ���������������ҪԪ��֮һ���Ӵ�ͳ���ϵ���Ϣ���ϵķ�չ�����д�����һ����һ���漣��
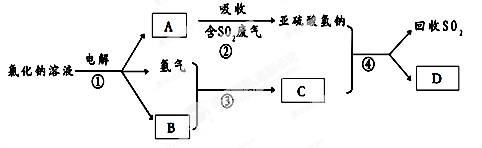
��1������Ԫ�����ڱ��е�λ����_______��
��2����ҵ�����ֹ�ķ�Ӧ�У�SiO2+2C Si���֣�+2CO����SiO2+3C
Si���֣�+2CO����SiO2+3C SiC+2CO��������Ʒ�е��ʹ���̼��������ʵ���֮��Ϊ1��1����μӷ�Ӧ��C��SiO2������֮��Ϊ ��
SiC+2CO��������Ʒ�е��ʹ���̼��������ʵ���֮��Ϊ1��1����μӷ�Ӧ��C��SiO2������֮��Ϊ ��
��3����ҵ�Ͽ���ͨ������ͼ��ʾ��������ȡ���裺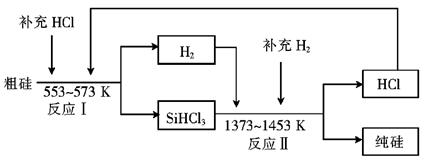
������ӦIΪ Si���֣�+3HCl SiHCl3+H2��
SiHCl3+H2��
��ӦII�Ļ�ѧ����ʽΪ ��
�������Ʊ����̱����ϸ������ˮ������SiHCl3��ˮ���ҷ�Ӧ����H2SiO3��HCl���� һ�����ʣ�д���÷�Ӧ�Ļ�ѧ����ʽ�� ��
�ۼ���ÿһ�ִ��Ʊ�1mol���裬�����������й�Ԫ��û����ʧ����ӦI��HCl��������Ϊ90%����ӦII��H2��������Ϊ93.75%�����ڵڶ��ִε������У�����Ͷ��HCl ��H2�����ʵ���֮���� ��
��1����������IVA�� ��2��1:2
��3����SiHCl3+H2 Si������+3HCl
Si������+3HCl
��SiHCl3+3H2O = H2SiO3+3HCl+H2��
��5:1
���������������1���������ڱ���λ�ڵ������ڵ�IVA�塣
��2����������ʽ���ӣ���֪C��SiO2�����ʵ���֮��Ϊ5:2�����������Ϊ����5��12������2��60��=1:2��
��3����������ͼ��֪��ӦII��SiHCl3��H2��Ӧ���ɴ����HCl����˷���ʽΪ��SiHCl3+H2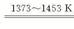 Si������+3HCl��
Si������+3HCl��
��д������ʽ����֪���ʣ�SiHCl3+3H2O �� H2SiO3+3HCl����ԭ���غ��֪��һ������ΪH2���ʷ���ʽΪSiHCl3+3H2O = H2SiO3+3HCl+H2����
��������������֪��
Si���֣�+ 3HCl 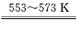 SiHCl3 + H2
SiHCl3 + H2
1mol 3mol 1mol
SiHCl3+H2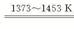 Si������+3HCl
Si������+3HCl
1mol 1mol 3mol
���������ݣ�ѭ��������ֻ�ܲ���3molHCl����HCl����������90%�������Ҫ���ӣ�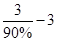 ��molHCl��ѭ��������ֻ����1molH2����H2��������Ϊ93.75%����������ӣ�
��molHCl��ѭ��������ֻ����1molH2����H2��������Ϊ93.75%����������ӣ� ��molH2����ˣ�����Ͷ��HCl ��H2�����ʵ���֮��Ϊ����
��molH2����ˣ�����Ͷ��HCl ��H2�����ʵ���֮��Ϊ����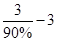 ������
������ ��=5:1��
��=5:1��
���㣺����Ԫ�����ڱ�������ʽ��д�����ݷ���ʽ�ļ��㡣

����Ԥ�ư��ǹ����������Դͷ����Ŀ��Ŀǰ���ڹ�����������Լ200�ң��ֿ�����ڹ���Ԥ�ư����������¡�
��1������Ԥ�ư������ά�ijɷ���ͬ����ô����Ԥ�ư��Ļ�ѧ�ɷ��� ��
��2������ѹ�����¶�ڼ��������У�������·�������ӷ���ʽ˵��ԭ�� ��
��3������˵����ȷ���� ��
| A��ʯӢ����ͨ�������ǹ�������Ʒ |
| B���������������õİ뵼����� |
| C������ʦ�������ʴʯӢ��������Ʒ |
| D��ʯӢ�������̼�еĻ�ѧ����ͬ |
��18�֣���ҵβ���е�������ͨ�����ð������շ�����ԭ����NH3��NOx�ڴ��������·�Ӧ�����������ʡ�ijУ�С��ͬѧ��������װ�úͲ���ģ�ҵ�ϵ������� �Ĵ������̡�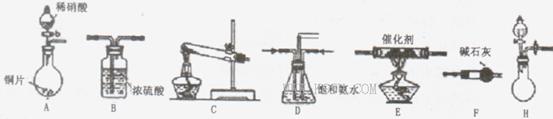
I��̽����ȡNH3�ķ���
��1��Bװ�õ����ƣ�______________
��2��������װ���У�H�ܿ��١������ȡ��װ������Ҫ���ӵķ�Ӧ�Լ�Ϊ_________ ��
��3��Ϊ̽�����õ�ʵ��Ч�����С��ͬѧ��������Cװ������ȡ�������ڿ���ʵ��������ͬ������£�����±���ʵ�����ݡ�
�����������ݣ�����Ϊ���ַ�����ȡ������Ч�����________������ţ����Ӹ÷���ѡ���ԭ�Ϸ�������Ч���õĿ���ԭ����________��__________��
| �Լ������� | �����Լ� | NH3�����mL�� | |
| a | 6.0gCa(OH)2���� | 5.4gNH4Cl | 1344 |
| b | 5.4g(NH4)2SO4 | 1364 | |
| c | 6.0gNaOH���� | 5.4gNH4Cl | 1568 |
| d | 5.4g(NH4)2SO4 | 1559 | |
| e | 6.0gCaO���� | 5.4gNH4Cl | 1753 |
| f | 5.4g(NH4)2SO4 | 1792 | |
II.ģ��β������
�С��ͬѧѡ����������װ�ã�������˳�����ӳ�ģ��β������װ�ý���ʵ�顣
��1���������װ����ѡ������Ϊ�����Ľ��в��䣨��ѡװ�ò����ظ�����

��2��A�з�Ӧ�����ӷ���ʽΪ__________________________
��3��Dװ�õ������У�_____________��____________��_____________��
��4��Dװ���е�Һ�廹�ɻ��� ������ţ���
A��H2OB��CCl4C��ŨH2SO4D��CuSO4��Һ
��5����С��ͬѧ����Ƶ�ģ��β������װ���л�����һ�����Ե�ȱ����___________.

 Si3N4 +12HCl
Si3N4 +12HCl