��Ŀ����
��13�֣�������ijС���ͬѧ��ͬ̽���Ҵ��������������Ӧʵ�飺
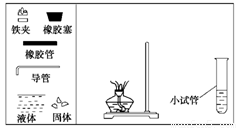
(1) �������ͼʾ������ʵ������ȡ����������װ��ͼ��
(2) ����С�Թ���װ���������____________������Ҫ������ �� ____________��__________________________��
(3) ����С�Թ�����ȡ����������Ӧʹ�õ���������______________��������ϣ�Ӧ�Ӹ�����________________ (��¿ڷš����Ͽڵ���)����
(4) ʵ����ϣ����ִ��Թ��еķ�ӦҺ��Щ��ڣ���ԭ����________________________��
(5) ��ʵ���У�����3mol�Ҵ���1mol������Ũ���������¼��ȣ���ַ�Ӧ���ܷ�����1mol���������� ���������� ����ܡ����ܡ�����ԭ���������������� ����������������ԭ����ģ���
(6) д���÷�Ӧ�Ļ�ѧ����ʽ��
(1)  ��2�֣�
��2�֣�
(2) ����Na2CO3��Һ���к����ᡢ�ܽ��Ҵ�����������������ˮ�е��ܽ�ȡ�
(3) ��Һ���Ͽڵ�
(4) ��Ũ���Ὣ��Ӧʣ����Ҵ�������̿��
(5) ���ܣ� �÷�Ӧ�ǿ��淴Ӧ��ת���ʲ��ܴﵽ100%
(6) CH3COOH+
C2H5OH CH3COOC2H5+H2O��2�֣�
CH3COOC2H5+H2O��2�֣�
����������1��������������ȡ����Һ���Һ��֮��ͨ��������ȡ��Ϊ�˷�ֹ���л���Ҫ�������Ƭ��װ����ͼ��ʾ��
��2���������ɵ����������л����������Ҵ�������Ϊ�˳�ȥ������Ҵ�����Ҫ���뱥��̼������Һ���������к����ᡢ�ܽ��Ҵ�����������������ˮ�е��ܽ�ȡ�
��3����������������ˮ������ͨ����Һ���з��롣��Һ����Ҫ�����Ƿ�Һ©������Ϊ�����������ܶ�С��ˮ�ģ�����Ҫ���Ͽڵ�����
��4������Ũ���������ˮ�ԣ�����ڷ�Ӧ�������п��ܽ���Ӧʣ����Ҵ�������̿�����Ӷ�ʹ��ӦҺ��ڡ�
��5������������Ӧ�ǿ��淴Ӧ�����Բ���������1mol����������
��6����Ӧ�ķ���ʽΪCH3COOH+ C2H5OH CH3COOC2H5+H2O��
CH3COOC2H5+H2O��

 ����ʦ���һ��һ��ϵ�д�
����ʦ���һ��һ��ϵ�д�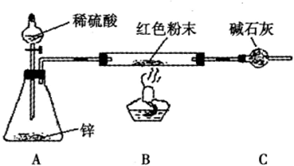

 ���µ����⣬̽��������Э����С���ͬѧ��������о���
���µ����⣬̽��������Э����С���ͬѧ��������о���
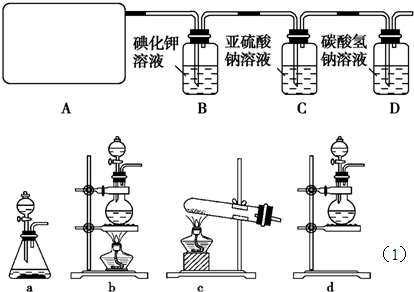
 �ܱ�NH3��ԭ��
�ܱ�NH3��ԭ�� ��ɫ������Cu��Ҳ��ͬѧ��ΪNH3��CuO��Ӧ���ɵĺ�ɫ����
��ɫ������Cu��Ҳ��ͬѧ��ΪNH3��CuO��Ӧ���ɵĺ�ɫ���� ��Cu��A�Ļ����������һ����ʵ�����NH3��CuO��Ӧ�����ɵĺ�ɫ�������Ƿ���A�ߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߡ�
��Cu��A�Ļ����������һ����ʵ�����NH3��CuO��Ӧ�����ɵĺ�ɫ�������Ƿ���A�ߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߣߡ�