��Ŀ����
1��Ϊ�˳�ȥFeSO4��Һ�е�Fe3+�����ü��봿��м�����˷��ܷ�ʱ������һ�ַ�����������ˮ����ʱ�����δ״���̷���������Σ������裬ֱ���۲쵽�к�ɫ������������ֹͣ����ҩƷ�����1��2���ӣ����ȹ��ˣ��ܷⱣ�����õ�FeSO4��Һ���˷���ԭ���ǣ�������| A�� | Fe3+��ˮ�⣬����ˮ�����֮һ�����Ƶ�Fe2+ˮ�� | |
| B�� | Fe2+�н�ǿ�Ļ�ԭ�ԣ����ܻ�ԭFe3+ | |
| C�� | Fe3+�н�ǿ�������ԣ���������Fe2+ | |
| D�� | Fe3+��ˮ�������ȷ�Ӧ�������ܴٽ�����ˮ�� |
���� Fe3+ˮ��IJ���֮һ��H+��H+������Fe2+��ˮ�⣬����Fe3+��ˮ�������ȷ�Ӧ�������ܴٽ�����ˮ�⣮
��� �⣺Fe3+ˮ��IJ���֮һ��H+��H+������Fe2+��ˮ�⣬����Fe3+��ˮ�������ȷ�Ӧ�������ܴٽ�����ˮ�⣬�������ԡ���ԭ���أ�
��ѡAD��
���� ���⿼�������ˮ�⣬�ѶȲ�����ȷʵ��ԭ���ǽ���Ĺؼ���

��ϰ��ϵ�д�
 �̲�ȫ���ִʾ�ƪϵ�д�
�̲�ȫ���ִʾ�ƪϵ�д�
�����Ŀ
11�����м������ʵ��۵��������±���
�ݴ˻ش��������⣺
��1��A������ԭ�Ӿ��徧�壬���ۻ�ʱ�˷���������������ǹ��ۼ���
��2��B�龧�干ͬ�����������Ǣ٢ڢۢܣ�����ţ������н������ڵ����ԡ��۵����ԡ�����չ��
��3��C����HF�۵㷴��������HF���Ӽ���������
��4��D�龧����ܾ��е������Ǣڢܣ�����ţ�����Ӳ��С����ˮ��Һ�ܵ��硡�۹����ܵ��硡������״̬�ܵ��磮
| A�� | B�� | C�� | D�� |
| ���ʯ��3110�� | Li��181�� | HF��-83�� | NaCl��801�� |
| �辧�壺1410�� | Na��98�� | HCl��-114�� | KCl��776�� |
| ���壺2300�� | K��64�� | HBr��-89�� | RbCl��718�� |
| �������裺1723�� | Rb��39�� | HI��-51�� | CsCl��645�� |
��1��A������ԭ�Ӿ��徧�壬���ۻ�ʱ�˷���������������ǹ��ۼ���
��2��B�龧�干ͬ�����������Ǣ٢ڢۢܣ�����ţ������н������ڵ����ԡ��۵����ԡ�����չ��
��3��C����HF�۵㷴��������HF���Ӽ���������
��4��D�龧����ܾ��е������Ǣڢܣ�����ţ�����Ӳ��С����ˮ��Һ�ܵ��硡�۹����ܵ��硡������״̬�ܵ��磮
12����֪�ס��ҡ�����X��������ѧ��ѧ�г��������ʣ���ת����ϵ��ͼ��ʾ�����X�������ǣ�������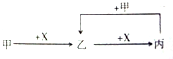

| A�� | ��ΪC��XΪO2 | B�� | ��ΪFe��XΪCl2 | ||
| C�� | ��ΪCO2��XΪNaOH��Һ | D�� | ��ΪHCl��XΪNa2CO3��Һ |
9����˿���صĽṹ��ʽ��ͼ��ʾ�������й�����˵����ȷ���ǣ�������


| A�� | ���ڷ����� | B�� | �����б����ϵ�һ�ȴ�����3�� | ||
| C�� | ����������Ʒ�����Ӧ | D�� | ���ܷ���ˮ�ⷴӦ |
16����̼������ָ�ӿ����в���CO2�ļ�����ĿǰNH3�Ѿ���������ҵ��̼�������������в�����Ϊ��̼�����ǣ�������
| A�� | 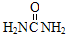 | B�� | H2NCH2CH2NH2 | C�� | ��NH4��2CO3 | D�� | Na2CO3 |
6�����б�ʾ���ʽṹ�Ļ�ѧ������ȷ���ǣ�������
| A�� | CH4���ӵı���ģ�ͣ�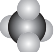 | B�� | CO2�ĵ���ʽ��O����C����O | ||
| C�� | Cl-�����ӽṹʾ��ͼ��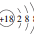 | D�� | ������Ϊ18����ԭ�ӣ�${\;}_{16}^{18}$S |
13������˵����ȷ���ǣ�������
| A�� | Na2O2�мȺ����Ӽ��ֺ��Ǽ��Լ� | |
| B�� | BF3�����е�����ԭ�ӹ�ƽ�� | |
| C�� | SiO2��Na2O2�ľ���������ͬ | |
| D�� | S�ĵ�һ�����ܴ���P�ĵ�һ�����ܣ� |
10������Һ�壨Ionic Iiquid������ָ���л������Ӻ������ӹ��ɵ��������³�Һ̬����������������ѹ�͡��۵�͡�Һ�̿����ײ����������Ժú��ȶ��Ըߵ�����������Ӧ���ڿƼ��������������һ�ֺϳɡ�����Һ�塱��ԭ������ ������ʽ��C3H4N2���뱽�����ƽṹ�����ʣ��й�����˵��������ǣ�������
������ʽ��C3H4N2���뱽�����ƽṹ�����ʣ��й�����˵��������ǣ�������
 ������ʽ��C3H4N2���뱽�����ƽṹ�����ʣ��й�����˵��������ǣ�������
������ʽ��C3H4N2���뱽�����ƽṹ�����ʣ��й�����˵��������ǣ�������| A�� | �Ƿ����廯���� | |
| B�� | �ܷ����ӳɡ�ȡ����������Ӧ | |
| C�� | ÿ����������9���Ҽ� | |
| D�� | ���Ӽ��Լ��ֺ��зǼ��Լ��мȺ��� |
