��Ŀ����
��12�֣�
��һ�������£����и����ʿɷ�������ͼ��ʾ�ı仯��
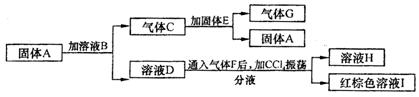
��֪��A��D��E��H����ɫ��Ӧ���ʻ�ɫ��G��ʹ�����ǵ�ľ����ȼ����ҺB�м�AgNO3��Һ��������ɫ��������ҺH�м�AgNO3��Һ������ɫ������
��1��д��A��B�Ļ�ѧʽ��A__________,B______________��
��2�� E�д��ڵĻ�ѧ������Ϊ��______________��_________________��
��3�� д������C����E��Ӧ�Ļ�ѧ����ʽ_______________________________��
��4�� д����ҺD������F��Ӧ�����ӷ���ʽ_______________________________��
��1��Na2CO3 HBr����2�֣�
(2)���Ӽ� �Ǽ��Թ��ۼ� ����2�֣�
(3)2CO2+2NaO2![]() 2Na2CO3+O2��2�֣�
2Na2CO3+O2��2�֣�
(4)2Br-+Cl2![]() Br2+2Cl-��2�֣�
Br2+2Cl-��2�֣�

��ϰ��ϵ�д�
 ȫ�ſ��䵥Ԫ�����������ܸ�ϰϵ�д�
ȫ�ſ��䵥Ԫ�����������ܸ�ϰϵ�д�
�����Ŀ
 �ס��ҡ����������������ֶ�����Ԫ�أ���ԭ�����������������ǿ���������п�ͼ�������ʣ������ҡ������Ҿ����γ�ԭ�Ӹ�����Ϊ2��1��1��1�Ļ������Ԫ��ԭ�ӵ�����������������Ӳ�����2������ͼ����ҺMΪ����ɫ����һ�������£����и����ʿɷ�����ͼ��ʾ�ı仯���Իش�
�ס��ҡ����������������ֶ�����Ԫ�أ���ԭ�����������������ǿ���������п�ͼ�������ʣ������ҡ������Ҿ����γ�ԭ�Ӹ�����Ϊ2��1��1��1�Ļ������Ԫ��ԭ�ӵ�����������������Ӳ�����2������ͼ����ҺMΪ����ɫ����һ�������£����и����ʿɷ�����ͼ��ʾ�ı仯���Իش�





