��Ŀ����
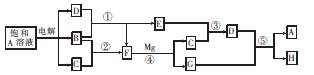
��16�֣���ͼ����ѧ�������ʼ��ת����ϵ����֪��
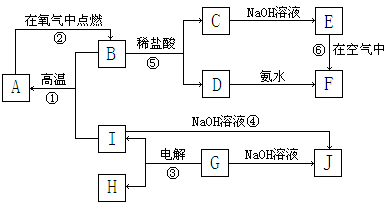
a��AΪ����ɫ���壬BΪ���¡�����ЧӦ������Ҫ���ʣ�
b��EΪ����������JΪ���ɫ������
c��G��ʵ�����г����ڼ���B�Ĵ��ڣ�
d��L��һ����Ҫ�Ĺ�ҵԭ�ϣ�����������ըҩ��Ũ��Һ�����治�����ʻ�ɫ��
�ش��������⣺

��1��A�ĵ���ʽΪ ��B�������� ���ӣ����ԡ��Ǽ��ԣ���
��2����Ӧ�ٵĻ�ѧ����ʽΪ ��
��Ӧ�ڵ����ӷ���ʽΪ ��
��3�����μӷ�Ӧ��A������Ϊ39g��������CO2�����������£�Ϊ L��
��4������K�������ӳ��õķ����� ��
��5��LŨ��Һ�ı��淽���� ��
a��AΪ����ɫ���壬BΪ���¡�����ЧӦ������Ҫ���ʣ�
b��EΪ����������JΪ���ɫ������
c��G��ʵ�����г����ڼ���B�Ĵ��ڣ�
d��L��һ����Ҫ�Ĺ�ҵԭ�ϣ�����������ըҩ��Ũ��Һ�����治�����ʻ�ɫ��
�ش��������⣺

��1��A�ĵ���ʽΪ ��B�������� ���ӣ����ԡ��Ǽ��ԣ���
��2����Ӧ�ٵĻ�ѧ����ʽΪ ��
��Ӧ�ڵ����ӷ���ʽΪ ��
��3�����μӷ�Ӧ��A������Ϊ39g��������CO2�����������£�Ϊ L��
��4������K�������ӳ��õķ����� ��
��5��LŨ��Һ�ı��淽���� ��
(16�֣�����ʽÿ��3�֣�����ÿ��2��)
��1���ԣ��Ǽ��Լ�
��2��2Na2O2+2CO2=2Na2CO3+O2�� Fe3++3OH��=Fe��OH��3��
��3��5��6L��
��4�����Թ���ȡ����K��Һ���μӼ���KSCN��Һ������Һ��ɫ��֤����Fe3+��
��5������ɫ�Լ�ƿ�ӱ��沢�����ڵ��±ܹ
��1���ԣ��Ǽ��Լ�
��2��2Na2O2+2CO2=2Na2CO3+O2�� Fe3++3OH��=Fe��OH��3��
��3��5��6L��
��4�����Թ���ȡ����K��Һ���μӼ���KSCN��Һ������Һ��ɫ��֤����Fe3+��
��5������ɫ�Լ�ƿ�ӱ��沢�����ڵ��±ܹ
��

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ

 C D
C D  ����Cl����NO3���е�һ�ֻ��֣�ȡ����Һ��������ʵ�飬ʵ��������£�
����Cl����NO3���е�һ�ֻ��֣�ȡ����Һ��������ʵ�飬ʵ��������£�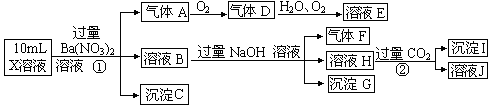
 ____________ _____ ������____________ __________
____________ _____ ������____________ __________