��Ŀ����
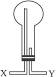
ʵ������ͼװ����ȡ�����屽��1������ƿa��װ���Լ���

��2�������Ʋⳤֱ����b�����ã�һ��
��3�������������c���¿ڿɷ��û��Һ���У�
��4����Ӧ��Ϻ�����ƿd�еμ�AgNO3��Һ��
��5����Ӧ��Ϻ���ƿa�е�Һ�嵹��ʢ����ˮ���ձ�����Թ۲쵽�ձ��ײ��к�ɫ������ˮ��Һ�壬���������Ϊ
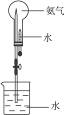
��2������Һ�巢��ȡ����Ӧ�������屽��HBr�����ڷ�Ӧ���ȣ��塢���е�ͣ��ӷ������Դӵ��ܵ����������п��ܺ���һ������ͱ���
��3��HBr��������ˮ��
��4��HBr������������Һ��������ӦHBr+AgNO3=AgBr��+HNO3������AgBr����ɫ������
��5�����屽Ϊ��ɫҺ�壬�ܶȱ�ˮ���屽����δ��Ӧ���壬���ʺ�ɫ����ȥ�����屽�е��壬�ɼ�������������Һ���ٷ�Һ��
 +Br2
+Br2| Fe |
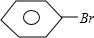 +HBr��
+HBr���ʴ�Ϊ���壻�������ۣ�
 +Br2
+Br2| Fe |
 +HBr��
+HBr����2������Һ�巢��ȡ����Ӧ�������屽��HBr�����ڷ�Ӧ���ȣ��塢���е�ͣ��ӷ������Դӵ��ܵ����������п��ܺ���һ������ͱ����ʳ����ܵ�����Ϊ������������������Ӧ��ʴ�Ϊ��������������������Ӧ�
��3��HBr��������ˮ�����ܽ�����cĩ�˽�û��Һ���У�����cĩ������ƿ��Һ�汣��һ�ξ����Ŀ���Ƿ��������𣺲��ܣ���ֹ������
��4��HBr������������Һ��������ӦHBr+AgNO3=AgBr��+HNO3������AgBr����ɫ���������ӷ���ʽΪAg++Br-=AgBr�����ʴ�Ϊ��dz��ɫ������Ag++Br-=AgBr����
��5�����屽Ϊ��ɫҺ�壬�ܶȱ�ˮ���屽����δ��Ӧ���壬���ʺ�ɫ����ȥ�����屽�е��壬�ɼ�������������Һ����ַ�Ӧ���÷ֲ㣬��Һȡ�²�Һ��
�ʴ�Ϊ���屽����δ��Ӧ���壻�ޣ�Һ���أ�����������Һ����ַ�Ӧ���÷ֲ㣬��Һȡ�²�Һ��

 ���ƽ̸�������ѡ����ĩ���100��ϵ�д�
���ƽ̸�������ѡ����ĩ���100��ϵ�д�NaBr+H2SO4![]() NaHSO4+HBr
NaHSO4+HBr
CH3CH2OH+HBr![]() CH3CH2Br+H2O
CH3CH2Br+H2O
��֪��Ӧ���������NaBr(����)0.3 mol���Ҵ�0.25mol��Ũ����36 mL(98%���ܶ�

ͼ6-2
(1)����ͼ6-2��ʾ������װ��ȡ���ռ��������װ�ã�Ҫ��ﵽ��ȫ����ʧ�١�����Ⱦ������Ŀ�ģ��й�������ѡ�������˳��Ϊ(������)____________��
(2)д������ѡ�õ�����(���������룬��ס��ҡ���)�����ɡ�
����ѡ�õ����� | ���� |
|
|
|
|
|
|
(3)��Ӧʱ�����¶ȹ��ߣ��ɼ�����ɫ���������д����Ӧ�Ļ�ѧ����ʽ��_________��
(4)���ػ�ɫ�Ĵ���������ȡ��ɫ�������飬Ӧ������Լ���_________������ʹ�õ�������_________��
(5)��ʵ��IJ���Ϊ60%�������ȡ������_________g��
��������һ��������ˮ����ɫҺ�壬�ܶ�ԼΪˮ��1.5�����е�Ϊ38.4 �档ʵ������ȡ������ķ�Ӧ���£�
NaBr+H2SO4![]() NaHSO4+HBr
NaHSO4+HBr
CH3CH2OH+HBr![]() CH3CH2Br+H2O
CH3CH2Br+H2O
��֪��Ӧ���������NaBr(����)0.3 mol���Ҵ�0.25mol��Ũ����36 mL(98%���ܶ�1.84 g��cm-3)��ˮ25 mL�������Ҵ����ܶ�ԼΪˮ���ܶȵ�4/5���Իش�

ͼ6-2
(1)����ͼ6-2��ʾ������װ��ȡ���ռ��������װ�ã�Ҫ��ﵽ��ȫ����ʧ�١�����Ⱦ������Ŀ�ģ��й�������ѡ�������˳��Ϊ(������)____________��
(2)д������ѡ�õ�����(���������룬��ס��ҡ���)�����ɡ�
| ����ѡ�õ����� | ���� |
(3)��Ӧʱ�����¶ȹ��ߣ��ɼ�����ɫ���������д����Ӧ�Ļ�ѧ����ʽ��_________��
(4)���ػ�ɫ�Ĵ���������ȡ��ɫ�������飬Ӧ������Լ���_________������ʹ�õ�������_________��
(5)��ʵ��IJ���Ϊ60%�������ȡ������_________g��
 �����������������ѧ��ѧ�еij������壬̽�����ߵ��Ʒ���������ʮ����Ҫ�Ŀ��⣮
�����������������ѧ��ѧ�еij������壬̽�����ߵ��Ʒ���������ʮ����Ҫ�Ŀ��⣮
 �������ڿ������ױ����������γɸ��ο��ȶ����ڣ����������[��NH4��2SO4?FeSO4?6H2O]Ϊdz��ɫ���壬��Ʒ��ΪĦ���Σ�������ˮ���������Ҵ��� ͨ���������²�����ʵ�����������Ʊ���
�������ڿ������ױ����������γɸ��ο��ȶ����ڣ����������[��NH4��2SO4?FeSO4?6H2O]Ϊdz��ɫ���壬��Ʒ��ΪĦ���Σ�������ˮ���������Ҵ��� ͨ���������²�����ʵ�����������Ʊ���