��Ŀ����
A��B��C��D��E����ѧ�γ�����5�ֻ����A��B�������Ԫ��X��Y�ĵ����������г����Ľ�����������ʼ��ת����ϵ��ͼ��ʾ��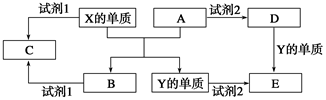
(1)X�ĵ�����A��Ӧ�Ļ�ѧ����ʽ��_______________________________��
(2)���Լ�1��NaOH��Һ����X�ĵ������Լ�1��Ӧ�����ӷ���ʽ��
___________________________________
(3)���Լ�1���Լ�2����ϡ���ᡣ
�ټ�������D����Һ�н������ӵķ�����___________________________________��
�ڽ�����C����ˮ������Һ�����ԣ�ԭ����(�����ӷ���ʽ��ʾ)
________________________________________________��
��ij��Ч��ˮ������Y(OH)SO4�ۺϵõ��ģ���ҵ����E��ϡ�������������Ϊԭ���Ʊ�Y(OH)SO4����Ӧ����NO���ɣ��÷�Ӧ�Ļ�ѧ����ʽ��_____________________��
(1)Fe2O3��2Al 2Fe��Al2O3
2Fe��Al2O3
(2)2Al��2OH����2H2O=2AlO2����3H2��
(3)��ȡ����D����Һ���Թ��У��μӼ���KSCN��Һ����Һ��Ѫ��ɫ��֤����Fe3��
��Al3����3H2O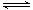 Al(OH)3��3H��
Al(OH)3��3H��
��2FeSO4��2NaNO2��H2SO4=2Fe(OH)SO4��Na2SO4��2NO��
����

 ��������ϵ�д�
��������ϵ�д�ij����С���һЩ�������ʺͻ���������ʽ���̽����
(1)�±�Ϊ�������Ȼ�ͭ��Һ��Ӧ��ʵ�鱨���һ���֣�
| ʵ�鲽�� | ʵ������ |
| ����ĥ������Ƭ(����)����һ��Ũ�ȵ�CuCl2��Һ�� | �������ݣ��������ɵĺ�ɫ���壬��Һ��Ϊ��ɫ |
| ��Ӧ������������Һ���� | |
| ��ɫ����������ˮϴ�Ӻ����ڳ�ʪ������ | һ��ʱ�������ɺ�ɫ��Ϊ��ɫ[������Ҫ�ɷ�ΪCu2(OH)2CO3] |
����Ӧ����д��ʵ���з�����Ӧ�Ļ�ѧ����ʽ��һ��(�����ӷ�Ӧ��ֻд���ӷ���ʽ)
�û���Ӧ______________________________________________________��
���Ϸ�Ӧ______________________________________________________��
(2)��ʯī���缫���������ʵ����������Һ�������������ݡ�������⣬��������������Һ�л����Թ۲쵽��������________________________��
���ʹ���������ӷ���ʽ��______________________________________��
(3)��ҵ�Ͽ����������̿�(��Ҫ�ɷ�ΪMnO2)��Ӧ��ұ�������̡�
�����������̿����̵�ԭ����(�û�ѧ����ʽ����ʾ)________________________��
��MnO2��H2O2�ֽⷴӦ������������������MnO2�����ữ��H2O2��Һ�У�MnO2�ܽ����Mn2�����÷�Ӧ�����ӷ���ʽ��_____________________________________��
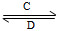 B(��ˮ��Һ�н���)
B(��ˮ��Һ�н���)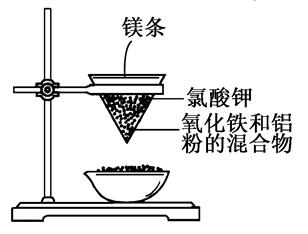

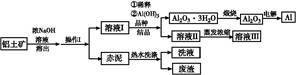
 2NaAlO2(aq)+4H2O,[Al2O3��3H2OҲ�ɱ�ʾΪ2 Al(OH)3]
2NaAlO2(aq)+4H2O,[Al2O3��3H2OҲ�ɱ�ʾΪ2 Al(OH)3]