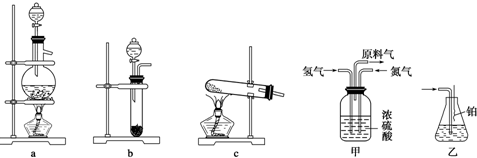
��Ŀ����
14��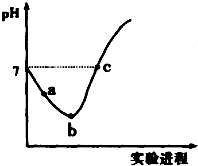 �����£���Cl2����ͨ��ˮ�������ͣ�Ȼ�������ñ�����ˮ�еμ�0.1mol•L-1��NaOH��Һ������ʵ���������Һ��pH�仯������ͼ��ʾ������������ȷ���ǣ�������
�����£���Cl2����ͨ��ˮ�������ͣ�Ȼ�������ñ�����ˮ�еμ�0.1mol•L-1��NaOH��Һ������ʵ���������Һ��pH�仯������ͼ��ʾ������������ȷ���ǣ�������| A�� | ��a����ʾ��Һ��ͨ��SO2����Һ�����Ժ�Ư���Ծ���ǿ | |
| B�� | b����ʾ����Һ�У�c��H+����c��Cl-����c��ClO-����c��HClO�� | |
| C�� | ��a�㵽b��Ĺ����У���Һ��$\frac{c��{H}^{+}��}{c��Cl{O}^{-}��}$��С | |
| D�� | c ����ʾ��Һ�У�c��Na+��=2c��ClO-��+c��HClO�� |
���� �������̷����ķ�ӦΪCl2+H2O�THCl+HClO��HCl+NaOH�TNaCl+H2O��HClO+NaOH�TNaClO+H2O��������Һ������Խ�ϵ���غ�������غ�������
A���������������ԣ��ܽ�������������Ϊ���ᣬ��������ԭΪ���
B��b ����ʾ��Һ�з�����Ӧ��Cl2+H2O=H++Cl-+HClO��HClOΪ������ʣ����ֵ��룬��Һ�д��ڵ������У�H+��Cl-��ClO-��OH-��H2O��HClO��Cl2��
C����a�㵽b��Ĺ������������ܽ�ƽ��Cl2+H2O?H++Cl-+HClO���ҽ��еĹ��̣���������ǿ��������Ũ��������
D��������Һ������Խ�ϵ���غ�������غ������
��� �⣺A���������������ԣ��ܽ�������������Ϊ���ᣬ��������ԭΪ���ᣬ���õ�����Һ���پ���Ư���ԣ���A����
B��b ����ʾ��Һ�з�����Ӧ��Cl2+H2O=H++Cl-+HClO��HClOΪ������ʣ����ֵ��룬��Һ�д��ڵ������У�H+��Cl-��ClO-��OH-��H2O��HClO��Cl2����Һ�У�c��H+����c��Cl-����c��HClO����c��ClO-������B����
C����a�㵽b��Ĺ������������ܽ�ƽ��Cl2+H2O?H++Cl-+HClO���ҽ��еĹ��̣���������ǿ��������Ũ����������Һ��$\frac{c��{H}^{+}��}{c��Cl{O}^{-}��}$����C����
D��c����Һ�����ԣ���c��H+��=c��OH-�������ݵ���غ��c��H+��+c��Na+��=c��Cl-��+c��Cl0-��+c��OH-��������c��Na+��=c��Cl-��+c��ClO-�������������غ��c��Cl-��=c��ClO-��+c��HClO��������c��Na+��=c��HClO��+2c��ClO-������D��ȷ��
��ѡD��
���� �����ۺϿ������������ʣ�������ѧ���ķ��������Ŀ��飬ע��������Һ�е����ʼ��������ǽⱾ��ؼ������������غ��c��Cl-��=c��ClO-��+c��HClO����Ϊ�״��㣬�Ѷ��еȣ�

��֪��N2��g��+2O2��g��=2NO2��g����H=+67.7kJ/mol
2N2H4��g��+2NO2��g��=3N2��g��+4H2O ��g����H=-1 135.7kJ/mol��
����˵����ȷ���ǣ�������
| A�� | N2H4��g��+O2��g��=N2��g��+2H2O��g����H=-1 068 kJ/mol | |
| B�� | �����백���Ƶ������������ˮ������뷽��ʽ��N2H4+H2O�TN2H5++OH- | |
| C�� | �����缫����KOH��ҺΪ�������Һ����--����ȼ�ϵ�أ��ŵ�ʱ�ĸ�����Ӧʽ��N2H4-4e-+4OH-=N2+4H2O | |
| D�� | �����缫����KOH��ҺΪ�������Һ����--����ȼ�ϵ�أ�����һ��ʱ���KOH��Һ��pH������ |
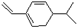 �����������ʵ���֮��Ϊ1��1��Br2�ӳ�ʱ�����ò����У�������
�����������ʵ���֮��Ϊ1��1��Br2�ӳ�ʱ�����ò����У�������| A�� | 3�� | B�� | 4�� | C�� | 5�� | D�� | 6�� |
| A�� | ��״���£�22.4 L SO3�к�����ԭ����ĿΪ3.0NA | |
| B�� | ���³�ѹ�£�12.0 gNaHSO4�к�����������Ϊ0.3NA | |
| C�� | ij�ܱ�����ʢ��0.1 mol N2��0.3 mol H2����һ�������³�ַ�Ӧ��ת�Ƶ��ӵ���ĿΪ0.6NA | |
| D�� | ��1 L 0.1 mol•L-1̼������Һ�У���������������0.1NA |
| A�� | ԭ�Ӱ뾶�Ĵ�С˳��Ϊ��rX��rY��rZ��rW��rQ | |
| B�� | Z��Q�γɵĻ�����Ϊ���ӻ����� | |
| C�� | ��������Ԫ��Z��Ԫ��W�γɵĻ������ˮ��Һ | |
| D�� | Ԫ��W������������Ӧ��ˮ��������Ա�Q��ǿ |
 ��ҵ����N2��H2 �ϳ�NH3��N2��g��+3H2��g���T2NH3��g����H��0���������ֿ��Խ�һ���Ʊ����ᣬ�ڹ�ҵ��һ��ɽ���������������ش��������⣺
��ҵ����N2��H2 �ϳ�NH3��N2��g��+3H2��g���T2NH3��g����H��0���������ֿ��Խ�һ���Ʊ����ᣬ�ڹ�ҵ��һ��ɽ���������������ش��������⣺��1����֪��
N2��g��+O2��g���T2NO��g����H=+a kJ•mol-1
N2��g��+3H2��g���T2NH3��g����H=-b kJ•mol-1
2H2��g��+O2��g��=2H2O��g����H=-c kJ•mol-1
����34g ��������������ȫ����һ�����������ˮ�������ų�������Ϊ��1.5c-a-b��kJ��
��2���ϳ�NH3�ﵽƽ���ijʱ�̸ı���������A���ڴﵽ��ƽ��Ĺ���������Ӧ����ʼ������
A������ B����ѹ C������c��N2�� D������c��NH3��
��3��ij����С���о����������������������£��ı���ʼ�����������ʵ����Ժϳ�NH3��Ӧ��Ӱ�죮ʵ��������ͼ1��ʾ����ͼ��T��ʾ�¶ȣ�n��ʾ��ʼʱH2���ʵ�����
��ͼ����T2��T1�Ĺ�ϵ�ǣ�T2��T1�������������������=������ȷ��������
�ڱȽ���a��b��c����������ƽ��״̬�У���Ӧ��N2��ת����������c������ĸ����
���������ݻ�Ϊ1L��b���Ӧ��n=0.15mol�����ƽ��ʱH2��ת����Ϊ60%����ƽ��ʱN2�����ʵ���Ũ��Ϊ0.02 mol•L-1��
��4��һ���¶��£���2mol N2��4mol H2����1L�ĺ����ܱ������з�Ӧ����ò�ͬ��������ͬʱ����ںϳ�NH3��Ӧ��N2��ת���ʣ��õ����������
 | 1Сʱ | 2 Сʱ | 3Сʱ | 4Сʱ |
| T3 | 30% | 50% | 80% | 80% |
| T4 | 35% | 60% | a | b |
��5������ȡ����ݵ��ܱ�������ͨ��H2��N2ʹ����һ�������ºϳ�NH3������ͼ2�л���ƽ�ⳣ��K��ʱ��ı仯���ߣ�
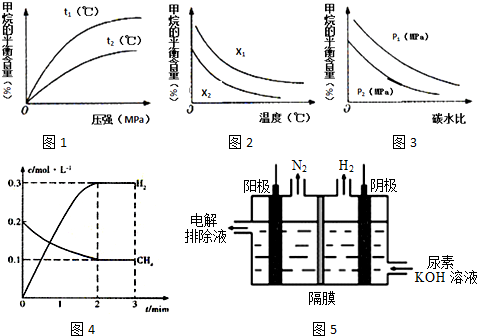
��1��д��������ˮ�����ڸ����·�Ӧ�ƺϳ������Ȼ�ѧ����ʽCH4��g��+H2O��g��=CO��g��+3H2��g����H=+206.1kJ•mol-1��
��2����һ�������£�������̶�Ϊ2L���ܱ������г���0.40mol CH4��g����0.60mol H2O��g�������CH4��g����H2��g�������ʵ���Ũ����ʱ��仯��������ͼ4��ʾ��
3minʱ�ı���������Ӧ���е�t1minʱ����ϵ�и����ʵ����ʵ������±���ʾ��
| t/min | n��CH4��/mol | n��H2O��/mol | n��CO��/mol | n��H2��/mol |
| t1 | 0.18 | 0.38 | 0.22 | 0.66 |
��3����֪�¶ȡ�ѹǿ��̼ˮ��[$\frac{n��C{H}_{4}��}{n��{H}_{2}O��}$]�Ը÷�Ӧ��Ӱ����ͼ��ʾ��
��ͼl�У�����������ʾ�¶ȵĹ�ϵ��t1��t2�����������������=������ͬ����
��ͼ2�У�����������ʾ̼ˮ�ȵĹ�ϵ��x1��x2��
��ͼ3�У�����������ʾѹǿ�Ĺ�ϵ��p1��p2��
��4������Ҳ�����õ�������Ʊ����������[CO��NH2��2]�ļ�����Һ�����װ��ʾ��ͼ5ͼ�������и�Ĥ����ֹ����ͨ��������������Ϊ���Ե缫�������ʱ�������ĵ缫��ӦʽΪCO��NH2��2+8OH--6e-=CO32-+N2��+6H2O��
| A�� | �������ӵ������ԣ�Z��X | |
| B�� | ��̬�⻯����ȶ��ԣ�M��Y | |
| C�� | �����Ӱ뾶�ɴ�С��˳���ǣ�M��Y��Z | |
| D�� | ��X2M��Z2M����Һ�У�ˮ�ĵ���̶ȣ�ǰ�ߴ��ں��� |