��Ŀ����
����˵�����ʾ������ȷ���ǣ�������
| A�������ʵ������������������ֱ���ȫȼ�գ����߷ų������� |
| B����H+��aq��+OH-��aq����H2O��l��+57.3 kJ��֪��������1 mol CH3COOH��ϡ��Һ�뺬1 mol NaOH��ϡ��Һ��ϣ��ų�������С��57.3 kJ |
| C����C��ʯī����C�����ʯ��-1.90 kJ��֪�����ʯ��ʯī�ȶ� |
| D����101 kPaʱ��2 g H2��ȫȼ������Һ̬ˮ���ų�285.8 kJ������������ȼ�յ��Ȼ�ѧ����ʽΪ�� 2H2��g��+O2��g����2H2O��l��+285.8 kJ |
A�������ʵ������������������ֱ���ȫȼ�գ��������仯Ϊ������ȣ�ǰ�߷ų������࣬�ʴ���
B��������������ڵ���ƽ�⣬������������ȹ��̣�������1 mol CH3COOH��ϡ��Һ�뺬1 mol NaOH��ϡ��Һ��ϣ��ų�������С��57.3 kJ����B��ȷ��
C����C��ʯī����C�����ʯ��-1.90 kJ��֪��ʯī�����ͣ�ʯī�Ƚ��ʯ�ȶ�����C����
D����101 kPaʱ��2 g H2��ȫȼ������Һ̬ˮ���ų�285.8 kJ������������ȼ�յ��Ȼ�ѧ����ʽΪ�� H2��g��+
O2��g����H2O��l��+285.8 kJ����D����
��ѡB��
B��������������ڵ���ƽ�⣬������������ȹ��̣�������1 mol CH3COOH��ϡ��Һ�뺬1 mol NaOH��ϡ��Һ��ϣ��ų�������С��57.3 kJ����B��ȷ��
C����C��ʯī����C�����ʯ��-1.90 kJ��֪��ʯī�����ͣ�ʯī�Ƚ��ʯ�ȶ�����C����
D����101 kPaʱ��2 g H2��ȫȼ������Һ̬ˮ���ų�285.8 kJ������������ȼ�յ��Ȼ�ѧ����ʽΪ�� H2��g��+
| 1 |
| 2 |
��ѡB��

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
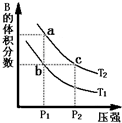 ���ڷ�ӦA��g��?2B��g����H��0�����¶�ΪT1��T2ʱ��ƽ����ϵ��B�����������ѹǿ�仯��������ͼ��ʾ���ش����и��⣮
���ڷ�ӦA��g��?2B��g����H��0�����¶�ΪT1��T2ʱ��ƽ����ϵ��B�����������ѹǿ�仯��������ͼ��ʾ���ش����и��⣮

