��Ŀ����
5��ij��ѧ����С���ú���Ϊԭ����ȡ��������ˮ������CCl4�ӵ�ˮ����ȡ�Ⲣ�÷�Һ©������������Һ����ʵ������ɷֽ�Ϊ���¼�����A����ʢ����Һ�ķ�Һ©����������̨����Ȧ�У�
B����50mL��ˮ��15mL CCl4�����Һ©���У����Ǻò�������
C�������Һ©���������Ͽڲ������Ƿ�©Һ��
D����ת©������������ʱ�����������������رջ������ѷ�Һ©��������
E���ſ����������ձ�������Һ��
F���ӷ�Һ©���Ͽڵ����ϲ�ˮ��Һ��
G����©���ϿڵIJ���������ʹ��Һ�ϵİ��ۻ�С��©�����ϵ�С�ף�
H�����á��ֲ㣮
�ʹ�ʵ�飬���������գ�
��1����ȷ���������˳���ǣ��������������ı����ĸ��գ���
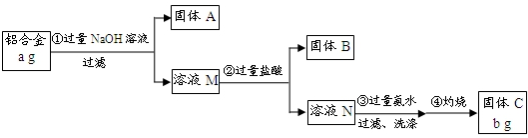
C��B��D��A��G��H��E��F��
��2������E����IJ�����Ӧע��ʹ©���¶˹ܿڽ����ձ��ڱڣ���ʱ�رջ�������Ҫ���ϲ�Һ��������
��3������G���������Ŀ���Dz���ʱ©����Һ���ܹ�������
��4����ѡ��CCl4�ӵ�ˮ����ȡ���ԭ����CCl4��ˮ�����ܣ����ҵ���CCl4�е��ܽ�ȱ���ˮ�еĴ�ö࣮
��5���������ʲ�����Ϊ����ˮ����ȡ����ܼ���A��
A���ƾ� B������һ���л��ܼ�����ˮ�������ܣ��ܶȱ�ˮС�� C�����Ȼ�̼��
���� ��1��CCl4�ӵ�ˮ����ȡ�Ⲣ�÷�Һ©������������Һ������Ϊ��©��װҺ�������á���Һ��
��2�����������������²�Һ�壻
��3����G�������ʹҺ��˳�����£�
��4������ˮ�����Ȼ�̼�е��ܽ��Բ�ͬ��
��5����ȡ����ѡ��ԭ����������һ����������ȡ���е��ܽ�ȱ���ˮ��Ҫ��öࣻ������ȡ����ˮ�����ܽ��ֳܷ����㣻������ȡ�������ʲ��ᷢ����Ӧ��
��� �⣺��1��CCl4�ӵ�ˮ����ȡ�Ⲣ�÷�Һ©������������Һ������Ϊ��©��װҺ�������á���Һ������ΪC��B��D��A��G��H��E��F���ʴ�Ϊ��C��B��D��H��
��2�����������������²�Һ�壬Ӧʹ©���¶˹ܿڽ����ձ��ڱڣ���ʱ�رջ�������Ҫ���ϲ�Һ��������
�ʴ�Ϊ��ʹ©���¶˹ܿڽ����ձ��ڱڣ���ʱ�رջ�������Ҫ���ϲ�Һ��������
��3����G������Ŀ��Ϊ����ʱ©����Һ���ܹ�������
�ʴ�Ϊ������ʱ©����Һ���ܹ�������
��4��ѡ��CCl4�ӵ�ˮ����ȡ���ԭ����CCl4��ˮ�����ܣ����ҵ���CCl4�е��ܽ�ȱ���ˮ�еĴ�ö࣬
�ʴ�Ϊ��CCl4��ˮ�����ܣ����ҵ���CCl4�е��ܽ�ȱ���ˮ�еĴ�öࣻ
��5���ƾ���ˮ������������ܣ����ֲ㣬������Ϊ��ȡ�����ʴ�Ϊ��A��
���� ���⿼���˷�Һ�IJ������ȡ����ѡ��ѡ����ȡ��������ȡ����ѡ����жϼ��ɣ�Ҫע��������ͺ����������������ϵIJ�ͬ����Ŀ�Ѷ��еȣ�

| A�� | 1molŨ���������ͭ�۷�Ӧת�Ƶĵ�����ΪNA | |
| B�� | ��״���£�5.6LNO��5.6 LO2��Ϻ�ķ�������ΪΪ0.5NA | |
| C�� | 0.1mol����ϩ�к��е�̼̼˫������ĿΪ0.1 NA | |
| D�� | ��NA��NH3��������1Lˮ�еõ�1 mol•L-1�İ�ˮ |
Na2CO3+nH2O=Na2CO3•nH2O��nΪƽ��ֵ��n��10����ȡû�����Ʊ����Ѳ��ֱ��ʵ�NaHCO3��ƷA18.32g������ˮ�����Һ����������ϡ���Ტ��ͣ�ؽ��裬�����������������ɵ�CO2���������״�������±�������������ˮ��CO2���壩
| ���������mL�� | 5 | 15 | 20 | 50 | x | 120 | 150 |
| ����CO2�����mL�� | 0 | 224 | 448 | 1792 | 4480 | 4480 | 4480 |
��1�����������������CO2�����������ʱ��x����СֵΪ110��
��2��δ����ǰNaHCO3��������
��3�����ֱ��ʺ���ƷA�еijɷּ����ɷֵ����ʵ�����
| A�� | ����������ʵ����ձ��þƾ�ϴ�� | |
| B�� | ����������Ӧʵ����Թ���ϡ����ϴ�� | |
| C�� | �����ͷ��ڴ���Ƥ���IJ���ƿ�� | |
| D�� | ��˫��ˮ�е��뼸�����Ȼ�����Һ�ӿ�ֽ����� |
| A�� | Na+��Fe2+��NO3-��H+ | B�� | K+��Cu2+��NO3-��H+ | ||
| C�� | H+��Na+��NO3-��HCO3- | D�� | NH4+��Fe2+��NO3-��OH- |
| A�� | KAl ��SO4��2=K++Al3++2 SO42- | B�� | H2CO3=H++HCO3- | ||
| C�� | Ca��HCO3��2=Ca2++2H++2CO32- | D�� | Ba��OH��2=Ba2++OH2- |