��Ŀ����
A��B��C��D��Ϊ��ѧ��ѧ�ij��������Ҿ�����ͬһ��Ԫ�أ�����֮���ת����ϵ����ͼ��ʾ����Ӧ���������������Ѿ���ȥ����
A![]() B
B![]() C
C![]() D
D
��1����A��D��ˮ��Һ����ʹʪ�����ɫʯ����ֽ��죬��A��D�ֱ�Ϊ����д��ѧʽ����
A.___________________��D.___________________��
д��A��Bת���Ļ�ѧ����ʽ��___________________________________________________��
��2����A��ˮ��Һ��ʹʪ��ĺ�ɫʯ����ֽ������D��ϡ��Һ��ʹ��ɫ��ʪ��ʯ����ֽ��죬��AΪ_______________��д��A��Bת���Ļ�ѧ����ʽ��_______________________��
��3����AΪ���ý���Ԫ�صĵ��ʣ�DΪǿ���ɫ��Ӧ�Ի�ɫ����A��_______________��B��_______________��C��_______________��C����Ϊ������ߵ�������C�������̼������Ӧ�Ļ�ѧ����ʽΪ��______________________________________________________��A��ˮ������Ӧ�����ӷ���ʽΪ��________________________________________________��
��4����AΪ�����ķǽ������ʣ���A�ж������������д�±����������Ӧ�Ļ�ѧ��Ӧ����ʽ�������Բ�������Ҳ���Բ��䣩
| ��� | A | B��C��ѧ����ʽ |
| �� | ||
| �� | ||
| �� |
��1��H2S H2SO4 2H2S+3O2====2SO2+2H2O
��2��NH3 4NH3+5O2====4NO+6H2O
��3��Na Na2O Na2O2 2Na2O2+2CO2====2Na2CO3+O2 2Na+2H2O====2NaOH+H2��
��4����S 2SO2+O2![]() 2SO3 ��C 2CO+O2====2CO2 ��N2 2NO+O2
2SO3 ��C 2CO+O2====2CO2 ��N2 2NO+O2![]() 2NO2
2NO2
����:
�����ƶϵĻ������������á���С��Χ������ͻ�ơ�������������ѧ������������Խ���Χ��С���٣�������������ˮ��ʼ��ԣ�ʹʯ����Һ����ɫ�������塪��������

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�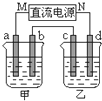 ��ͼ��ʾ��װ���У��ס������ձ��зֱ�ʢ����������CuSO4��Һ��100g 10.00%��K2SO4��Һ��a��b��c��d��Ϊʯī�缫����ͨ��Դһ��ʱ��������K2SO4��ҺŨ��Ϊ10.47%������a�缫���������ӣ�����˵����ȷ���ǣ�������
��ͼ��ʾ��װ���У��ס������ձ��зֱ�ʢ����������CuSO4��Һ��100g 10.00%��K2SO4��Һ��a��b��c��d��Ϊʯī�缫����ͨ��Դһ��ʱ��������K2SO4��ҺŨ��Ϊ10.47%������a�缫���������ӣ�����˵����ȷ���ǣ�������| A���ס�����Һ��pH����С | B���缫b��������������ԼΪ2.8L����״���£� | C���缫d�Ϸ����ķ�ӦΪ��2H2O+2e-?H2��+2OH- | D����ʹ���е���Һ�ָ���ԭ����Ũ�ȣ��ɼ���24.5g��Cu��OH��2 |
 A��B��C��D��Ϊ�������ʣ��֮��Ĺ�ϵ��ͼ��ʾ����-����ʾ�������ʼ��ܷ�����Ӧ����������ʾ���ʼ��ת����ϵ�����ַ�Ӧ����������Լ���Ӧ��������ȥ����
A��B��C��D��Ϊ�������ʣ��֮��Ĺ�ϵ��ͼ��ʾ����-����ʾ�������ʼ��ܷ�����Ӧ����������ʾ���ʼ��ת����ϵ�����ַ�Ӧ����������Լ���Ӧ��������ȥ����
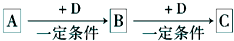 A��B��C��D��Ϊ��ѧ��ѧ�г��������ʣ�����֮���ת����ϵ����ͼ�����ֲ�������ȥ�����Իش�
A��B��C��D��Ϊ��ѧ��ѧ�г��������ʣ�����֮���ת����ϵ����ͼ�����ֲ�������ȥ�����Իش�
 A��B��C��D��Ϊ������Ԫ�أ�A��B��ͬ�������ڵ�����Ԫ�أ�A��C��ͬ�������ڵ�����Ԫ�أ�A��B��C����Ԫ�ص�ԭ������֮��Ϊ31��DԪ����A��B��C����Ԫ�ؼȲ���ͬ���ڣ�Ҳ��ͬ���壮��ش�
A��B��C��D��Ϊ������Ԫ�أ�A��B��ͬ�������ڵ�����Ԫ�أ�A��C��ͬ�������ڵ�����Ԫ�أ�A��B��C����Ԫ�ص�ԭ������֮��Ϊ31��DԪ����A��B��C����Ԫ�ؼȲ���ͬ���ڣ�Ҳ��ͬ���壮��ش�
 NH3?H2O+H+
NH3?H2O+H+ 2NH3
2NH3
 ���ͱ����γ����ֻ�����X��Y��X��ˮ��Ӧ������һ�־��л�ԭ�ԵĶ�Ԫ��M��1mol�����������ɻ�������Z�����õ�Z����ˮ��Ӧ�IJ���W����12mol KOH������ȫ�кͣ�������������ȼ�����ɻ�����N��N��ˮ��Ӧ����W��DԪ�ص���̬�⻯��Իش��������⣺
���ͱ����γ����ֻ�����X��Y��X��ˮ��Ӧ������һ�־��л�ԭ�ԵĶ�Ԫ��M��1mol�����������ɻ�������Z�����õ�Z����ˮ��Ӧ�IJ���W����12mol KOH������ȫ�кͣ�������������ȼ�����ɻ�����N��N��ˮ��Ӧ����W��DԪ�ص���̬�⻯��Իش��������⣺