��Ŀ����
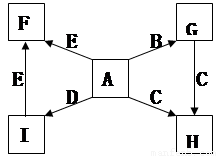
��ÿ��2�֣���14�֣�A��B��C��D��E��F������ѧ��ѧ�г��������ʣ�����ͼת����ϵ����
��1����A��C��E��Ϊ�������EΪ�ж����壬B��D��F��Ϊ�ǽ������ʣ���DΪ���塣��A�Ľṹ��ʽΪ ������F�ľ�������Ϊ ��1molC�к��� �����ۼ���
��2����A��C��E��Ϊ�⻯����߽�Ϊ���Է��ӣ�����C����Է���������С��D��E��F��Ϊ���������D��FΪ�ж����壬F��������Ϊ����ɫ��
��A��C��E�е��ɸߵ��͵�˳��Ϊ �����ѧʽ����A��C��E������̬�⻯���ȶ�����ǿ���� ���ѧʽ����
��B��C��Ӧ����E��F�Ļ�ѧ����ʽΪ ��
��A��D�����·�Ӧ������һ�ֵ��ʣ���ѧ����ʽΪ ��
��ÿ��2�֣���14�֣���1��H-O-H��ԭ�Ӿ��壬4NA����2����H2O��NH3��H2S��H2O��
��4NH3 + 5O2  4NO +6 H2O�� ��SO2 + 2H2S��2H2O + 3S��
4NO +6 H2O�� ��SO2 + 2H2S��2H2O + 3S��
����

��ϰ��ϵ�д�
 ���ɶ��ȫ���ƿؾ�ϵ�д�
���ɶ��ȫ���ƿؾ�ϵ�д�
�����Ŀ
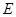 ���Է�����Ӧ��2E��I
���Է�����Ӧ��2E��I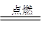 2F��D��F�е�EԪ�ص���������Ϊ60%.
2F��D��F�е�EԪ�ص���������Ϊ60%.