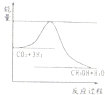
��Ŀ����
����ȷ��ʾ���л�ѧ��Ӧ�����ӷ���ʽ��ѧ����ʽ���ǣ� ��
A�����������缫�������������Һ��2H2O![]() 2H2��+O2��
2H2��+O2��
B����ȩ��������Ӧ��
CH3CHO+2[Ag(NH3)2]++2OH-![]() CH3COO��+NH4++2Ag��+3NH3+H2O
CH3COO��+NH4++2Ag��+3NH3+H2O
C����̼������Һ����������������2CO32-+2SO2+H2O=2HCO3-+SO32-
D��̼���Ⱶ��Һ�м�������������������Һ��Ba2++HCO3-+OH��=BaCO3��+ H2O
B

��ϰ��ϵ�д�
�����Ŀ
�����и�˵���У���ȷ���ǣ�������
| A������2SO2��g��+O2��g��?2SO3��g����H=-QkJ?mol-1����Q��0��������Ӧ�г���1molSO2��������������Ӧʱ���ų�������һ����Q/2 kJ | B���Ȼ�ѧ����ʽ�еĻ�ѧ�����������÷�����ʾ | C��Zn��s��+H2SO4��aq���TZnSO4��aq��+H2��g����H��0���÷�Ӧ�Ļ�ѧ�ܿ���ת��Ϊ���� | D����H�Ĵ�С���Ȼ�ѧ����ʽ�Ļ�ѧ�������� |
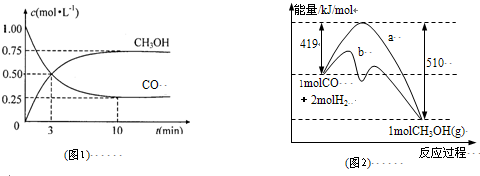
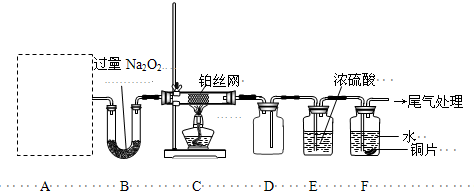
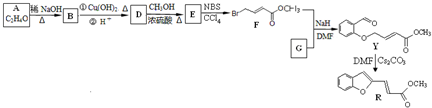
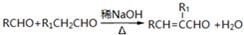 ��R��R1��ʾ�������⣩
��R��R1��ʾ�������⣩
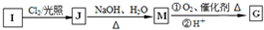


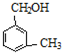 ��Ϊͬϵ��
��Ϊͬϵ��