��Ŀ����
����֪���ʵ���Ũ�ȵ�NaOH��Һ�ζ�δ֪Ũ�ȵ�����ʱ�����������²�����
��������ˮϴ�Ӽ�ʽ�ζ��ܺ�ֱ��װ����֪Ũ�ȵ�NaOH��Һ��
�ھ�����ʽ�ζ��ܲ�©Һ�������в��������������������֪��NaOH��Һϴ��2��3�κ���װ���Һ����Һ������̶ȡ�0�������õζ��ܼн���̶�������̨�ϣ�
���ô���������ϴ��Һ����ȡһ������Ĵ���������Һ������������ˮϴ������ƿ�У�
������ƿ�еμӼ��μ��ȣ���ƿ�µ�һ�Ű�ֽ��
�������ʽ�ζ����еIJ����飬���ٷ�Һ��ʹָʾ�������ɫ��
��ָʾ����ɫ���ٵμӼ��μ�Һ�����ȶ���ɫ��
���ϲ����д������(����)
A���٢ڢ� B���٢ڢ�
C���٢ݢ� D���ڢۢ�
C
��������
���������������������Ϥ�ζ�������Ҫ���ڵڢٲ���������ˮϴ�ӵζ��ܺ�����װ����֪Ũ�ȵ�NaOH��Һ�Ǵ���ģ�������������ʹ��֪Ũ�ȵ�NaOH��Һ��ϡ��ʹ�ζ��õ�NaOH��Һ�����Ӵ���ʹ���������Ũ��ƫ�ߡ���ȷ��������������Һ��ϴ�ζ���2��3�Σ���װ��Һ�����еζ�ʱ����ʼ�μӱ�Һ�����ʿɿ�Щ�������ܹ����γ�ˮ���������ӽ��ζ��յ�ʱ�������ζ����ʣ�һ��һ�Σ�������ε��룬������һ�μ�Һ��ָʾ���ɺ�ɫ��Ϊ��ɫ��ͣ�Ű������ɫҲ���ٱ仯ʱ�����ﵽ�ζ��յ㡣�ʢݢ���Ҳ����ѡC��
���㣺��������к͵ζ�ʵ���л����������й��ж�
�������������е��Ѷȵ����⣬��Ҫ�ǿ���ѧ��������к͵ζ�ʵ��ԭ���Լ���������д���������ּ�ڿ���ѧ��������û���֪ʶ���ʵ�����������������������ѧ���淶�Ͻ���ʵ�鶯�ֲ�������������ѧ����ѧϰ��Ȥ�����ѧ����Ӧ��������

ijѧ��������֪���ʵ���Ũ�ȵ��������ⶨδ֪���ʵ���Ũ�ȵ�NaOH��Һʱ��ѡ�������ָʾ��������д���пհף�
��1���ñ���������Һ�ζ������NaOH��Һʱ,���ְ�����ʽ�ζ��ܵĻ���������ҡ����ƿ���۾�ע����ƿ����Һ��ɫ�ı仯��ֱ�������һ���������Һ�ɻ�ɫ��Ϊ��ɫ����______ _________Ϊֹ��
��2�����ζ���ʼ�ͽ���ʱ����ʽ�ζ����е�Һ����ͼʾ��������������Һ�����Ϊ___ _____ mL��
��3�����в����п���ʹ����NaOH��Һ��Ũ����ֵƫ�͵���_____________��
A.��ʽ�ζ���δ�ñ�Һ��ϴ��ֱ��ע���������Һ
B.�ζ�ǰʢ��NaOH��Һ����ƿ������ˮϴ����û����
C.��ʽ�ζ����ڵζ�ǰ�����ݣ��ζ���������ʧ
D��ȡ�������ʱ����ʼ���Ӷ������ζ�����ʱ���Ӷ���
��4��ijѧ����������ʵ��ֱ��¼�й��������±���
| �ζ����� | ��������������Һ�����/mL | 0.1000mol��L-1��������/mL | ||
| �ζ�ǰ�̶� | �ζ���̶� | ��Һ���/mL | ||
| ��һ�� | 25.00 | 0.00 | 26.11 | 26.11 |
| �ڶ��� | 25.00 | 1.56 | 30.30 | 28.74 |
| ������ | 25.00 | 0.22 | 26.31 | 26.09 |
�����������ݼ����NaOH��Һ�����ʵ���Ũ�ȡ�c��NaOH��= ��
ijѧ��������֪���ʵ���Ũ�ȵ��������ⶨδ֪���ʵ���Ũ�ȵ�NaOH��Һʱ��ѡ�������ָʾ��������д���пհף�
��1���ñ���������Һ�ζ������NaOH��Һʱ,���ְ�����ʽ�ζ��ܵĻ���������ҡ����ƿ���۾�ע����ƿ����Һ��ɫ�ı仯��ֱ�������һ���������Һ�ɻ�ɫ��Ϊ��ɫ����______ _________Ϊֹ��
��2�����ζ���ʼ�ͽ���ʱ����ʽ�ζ����е�Һ����ͼʾ��������������Һ�����Ϊ___ _____ mL��
��3�����в����п���ʹ����NaOH��Һ��Ũ����ֵƫ�͵���_____________��
A.��ʽ�ζ���δ�ñ�Һ��ϴ��ֱ��ע���������Һ
B.�ζ�ǰʢ��NaOH��Һ����ƿ������ˮϴ����û����
C.��ʽ�ζ����ڵζ�ǰ�����ݣ��ζ���������ʧ
D��ȡ�������ʱ����ʼ���Ӷ������ζ�����ʱ���Ӷ���
��4��ijѧ����������ʵ��ֱ��¼�й��������±���
| �ζ����� | ��������������Һ�����/mL | 0.1000mol��L-1��������/mL | ||
| �ζ�ǰ�̶� | �ζ���̶� | ��Һ���/mL | ||
| ��һ�� | 25.00 | 0.00 | 26.11 | 26.11 |
| �ڶ��� | 25.00 | 1.56 | 30.30 | 28.74 |
| ������ | 25.00 | 0.22 | 26.31 | 26.09 |
ijѧ��������֪���ʵ���Ũ�ȵ��������ⶨδ֪���ʵ���Ũ�ȵ�NaOH��Һʱ��ѡ�������ָʾ��������д���пհף�
��1���ñ���������Һ�ζ������NaOH��Һʱ,���ְ�����ʽ�ζ��ܵĻ���������ҡ����ƿ���۾�ע����ƿ����Һ��ɫ�ı仯��ֱ�������һ���������Һ�ɻ�ɫ��Ϊ��ɫ����______ _________Ϊֹ��
��2�����ζ���ʼ�ͽ���ʱ����ʽ�ζ����е�Һ����ͼʾ��������������Һ�����Ϊ___ _____ mL��
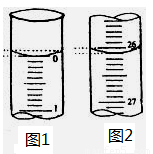
��3�����в����п���ʹ����NaOH��Һ��Ũ����ֵƫ�͵���_____________��
A.��ʽ�ζ���δ�ñ�Һ��ϴ��ֱ��ע���������Һ
B.�ζ�ǰʢ��NaOH��Һ����ƿ������ˮϴ����û����
C.��ʽ�ζ����ڵζ�ǰ�����ݣ��ζ���������ʧ
D��ȡ�������ʱ����ʼ���Ӷ������ζ�����ʱ���Ӷ���
��4��ijѧ����������ʵ��ֱ��¼�й��������±���
|
����� |
��������������Һ�����/mL |
0.1000mol��L-1��������/mL |
||
|
�ζ�ǰ�̶� |
�ζ���̶� |
��Һ���/mL |
||
|
��һ�� |
25.00 |
0.00 |
26.11 |
26.11 |
|
�ڶ��� |
25.00 |
1.56 |
30.30 |
28.74 |
|
������ |
25.00 |
0.22 |
26.31 |
26.09 |
�����������ݼ����NaOH��Һ�����ʵ���Ũ�ȡ�c��NaOH��= ��