��Ŀ����
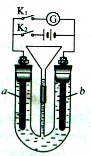
���װ����ͼ��ʾ��������װ��KI��������Һ���м��������ӽ���Ĥ��������һ���ĵ�ѹ��ͨ�磬���������Һ����ɫ��һ��ʱ�����ɫ��dz����֪��3I2��6OH��===IO��5I����3H2O������˵����ȷ����

A���Ҳ���ĵ缫��Ӧ������Ӧ
B��������ʱ���Ҳ���Һ�к���IO3-
C�������ڷ�����Ӧ���ܻ�ѧ����ʽ��2KI��2H2O KOH��H2��+I2
KOH��H2��+I2
D������������ӽ���Ĥ���������ӽ���Ĥ�������ڷ������ܻ�ѧ��Ӧ����
B
��������
���������A�������Һ����ɫ������I2�����缫Ϊ�������Ҳ�缫Ϊ�������缫��ӦʽΪ��2H2O+2e-=H2��+2OH-��������ԭ��Ӧ����A����B��һ��ʱ�����ɫ��dz��������Ӧ3I2+6OH-=IO3-+5I-+3H2O���м�Ϊ�����ӽ���Ĥ���Ҳ�I-��OH-ͨ�������ӽ���Ĥ������ƶ�����֤������Һ�ʵ����ԣ�����IO3-ͨ�������ӽ���Ĥ���Ҳ��ƶ������Ҳ���Һ�к���IO3-����B��ȷ��C�����缫Ϊ�������缫��ӦΪ��2I--2e-=I2��ͬʱ������Ӧ3I2+6OH-=IO3-+5I-+3H2O���Ҳ�缫Ϊ�������缫��ӦʽΪ��2H2O+2e-=H2��+2OH-�����ܵĵ缫��ӦʽΪ��KI+3H2O KIO3+3H2������C����D������������ӽ���Ĥ���������ӽ���Ĥ�����缫Ϊ�������缫��ӦΪ��2I--2e-=I2���Ҳ�缫Ϊ�������缫��ӦʽΪ��2H2O+2e-=H2��+2OH-����֤������Һ�ʵ����ԣ�������K+ͨ�������ӽ���ĤǨ�����������������I2���Ҳ���Һ����KOH���ɣ��ⵥ����KOH���ܷ�Ӧ���ܷ�Ӧ�൱�ڣ�2KI+2H2O
KIO3+3H2������C����D������������ӽ���Ĥ���������ӽ���Ĥ�����缫Ϊ�������缫��ӦΪ��2I--2e-=I2���Ҳ�缫Ϊ�������缫��ӦʽΪ��2H2O+2e-=H2��+2OH-����֤������Һ�ʵ����ԣ�������K+ͨ�������ӽ���ĤǨ�����������������I2���Ҳ���Һ����KOH���ɣ��ⵥ����KOH���ܷ�Ӧ���ܷ�Ӧ�൱�ڣ�2KI+2H2O  2KOH+I2+H2������D����
2KOH+I2+H2������D����
���㣺���ԭ��