��Ŀ����
��֪����M��ͬһ�����ڵ�X��Y����Ԫ����ɣ�X ԭ�ӵ������������������ڲ��������һ�룬YԪ�ص����������������۵Ĵ�����Ϊ6��M���������ʵ�ת����ϵ������ʾ�����ֲ�������ȥ��
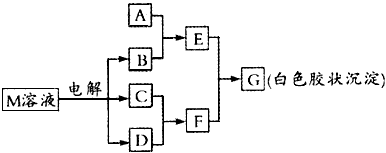
(1)��ҵ���M��Һ�Ļ�ѧ����ʽΪ____________________
(2)��A����X��Yͬ���ڵ�һ�ֳ�����������AԪ�������ڱ��е�λ���ǵ�____���ڵ�____�壬д��A��B��Һ��Ӧ�Ļ�ѧ����ʽ____________________��
(3)��A��ijԪ�ص�һ�ֳ������������������������ά�����Ԫ�ص�ԭ�ӽṹʾ��ͼΪ
________��д��E��F��Ӧ�����ӷ���ʽ____________________��
(4)B�ĵ���ʽΪ____________�����к��еĻ�ѧ��Ϊ____________��
(5)д��M���ʵ�һ����;��________________��
(2)��A����X��Yͬ���ڵ�һ�ֳ�����������AԪ�������ڱ��е�λ���ǵ�____���ڵ�____�壬д��A��B��Һ��Ӧ�Ļ�ѧ����ʽ____________________��
(3)��A��ijԪ�ص�һ�ֳ������������������������ά�����Ԫ�ص�ԭ�ӽṹʾ��ͼΪ
________��д��E��F��Ӧ�����ӷ���ʽ____________________��
(4)B�ĵ���ʽΪ____________�����к��еĻ�ѧ��Ϊ____________��
(5)д��M���ʵ�һ����;��________________��
(1)2NaCl+2H2O 2NaOH+H2��+Cl2��
2NaOH+H2��+Cl2��
(2)������A��2Al+2NaOH+2H2O=2NaAlO2+3H2��
(3)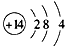 ��2H++SiO32-=H2SiO3��
��2H++SiO32-=H2SiO3��
(4)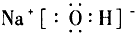 �����Ӽ����ۼ�
�����Ӽ����ۼ�
(5)�ȼҵ���𰸺������ɣ�
 2NaOH+H2��+Cl2��
2NaOH+H2��+Cl2��(2)������A��2Al+2NaOH+2H2O=2NaAlO2+3H2��
(3)
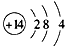 ��2H++SiO32-=H2SiO3��
��2H++SiO32-=H2SiO3��(4)
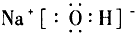 �����Ӽ����ۼ�
�����Ӽ����ۼ�(5)�ȼҵ���𰸺������ɣ�

��ϰ��ϵ�д�
�����Ŀ




 HSO3-+OH-
HSO3-+OH-