��Ŀ����
13������һ�ֺ��нᾧˮ���Σ���A��B��C��D��E����Ԫ����ɣ����������Ƕ�����Ԫ�أ�������ˮ��ɵ�������������Ӻ�һ�������ӣ�����A��B��Ԫ�ؿ��γ�10���ӵ������ӣ�AԪ��ԭ�Ӻ�����������E����1��D��E����ͬ���壮�ü�������ʵ�飺
��ȡ�����ľ�����������ˮ�����Һ��
��ȡ��������Һ���Թ��е���KSCN��Һ���������ٵ�����������������Һ���ʺ�ɫ��
��ȡ��������Һ���Թ��У������м���ϡ������ټ���BaCl2��Һ�����ְ�ɫ������
��ȡ��������Һ���Թ��м�������NaOH��Һ�����ȣ�
�ش��������⣺
��1��ʵ��ۿ�֤����Һ�к���SO42-��
��2�������ӷ���ʽ��ʾʵ����������ԭ��H2O2+2Fe2++2H+�T2H2O+2Fe3+��Fe3++3SCN-?Fe��SCN��3��
��3��ʵ����г��ֵ��������а�ɫ��״�������ɣ�Ѹ�ٱ�ɻ���ɫ�����ձ�ɺ��ɫ�������д����̼�����ζ�������ɣ�������ʹʪ��ĺ�ɫʯ����ֽ�������������ɣ���
��4�����ⶨ�����Ħ������Ϊ392g/mol�����������������������ʵ���֮��Ϊ3��2�������Ļ�ѧʽΪ��NH4��2Fe��SO4��2•6H2O��
���� ������ˮ��ɵ�����������ӣ����к�����A��B����Ԫ���γɵ�10���������ӣ�ӦΪ笠����ӣ�AԪ��ԭ�Ӻ�����������E����1������A��������HԪ�أ�ӦΪNԪ�أ���E��O��BΪHԪ�أ�D��E����ͬ���壬D��S��ȡ��������Һ���Թ��е������軯����Һ���������ٵ�����ˮ����Һ�ʺ�ɫ�������Һ�϶�����Fe2+��ȡ��������Һ���Թ��У������м���ϡ���ᣬ�ټ����Ȼ�����Һ�����ְ�ɫ�����������Һ�к���SO42-����������������Һ�к���Fe2+��SO42-��NH4+�������������������ʵ���֮��Ϊ3��2���Ļ�ѧʽ�����ǣ�NH4��2Fe��SO4��2�����ݾ����Ħ������Ϊ392g/mol�����Եõ�����Ļ�ѧʽΪ��NH4��2Fe��SO4��2•6H2O��������CΪFeԪ�أ��Դ˽����⣮
��� �⣺��1��ȡ��������Һ���Թ��У������м���ϡ���ᣬ�ټ����Ȼ�����Һ�����ְ�ɫ�����������Һ�к���SO42-���ʴ�Ϊ��SO42-��
��2������Һ�к���Fe2+������������ⷢ��H2O2+2Fe2++2H+�T2H2O+2Fe3+������Fe3+������KSCN����Fe3++3SCN-?Fe��SCN��3����Һ��ɺ�ɫ��
�ʴ�Ϊ��H2O2+2Fe2++2H+�T2H2O+2Fe3+��Fe3++3SCN-?Fe��SCN��3��
��3����������Һ���Թ��м�������NaOH��Һ������Fe2++2OH-=Fe��OH��2�����а�ɫ��״�������ɣ���������4Fe��OH��2+O2+2H2O=4Fe��OH��3��Ѹ�ٱ�ɻ���ɫ�����ձ�ɺ��ɫ��ͬʱ�ְ������ɣ�
�ʴ�Ϊ���а�ɫ��״�������ɣ�Ѹ�ٱ�ɻ���ɫ�����ձ�ɺ��ɫ�������д����̼�����ζ�������ɣ�������ʹʪ��ĺ�ɫʯ����ֽ�������������ɣ���
��4������Һ�к���Fe2+��SO42-��NH4+�������������������ʵ���֮��Ϊ3��2���ٸ��ݻ��ϼ۴�����Ϊ���֪���Ļ�ѧʽ�����ǣ�NH4��2Fe��SO4��2����Ϊ�����Ħ������Ϊ392g/mol�����Լ����к���ˮ������Ϊ$\frac{392-18��2-56-96��2}{18}$=6�����Ծ���Ļ�ѧʽΪ��NH4��2Fe��SO4��2•6H2O��
�ʴ�Ϊ����NH4��2Fe��SO4��2•6H2O��
���� ���⿼��������ƶϣ�Ϊ��Ƶ���㣬���ؿ���ѧ���ķ��������ͼ�����������Ŀ�Ѷ��еȣ�����ע����ݷ�Ӧ�������ж����ʣ�Ԫ�غ������ƶ��ǽ���Ĺؼ�������ʱע����ػ���֪ʶ��������ã�

| A�� | �����¶ȣ�X������������� | |
| B�� | ����ѹǿ����С�����������Z��Ũ�Ȳ��� | |
| C�� | ��������������䣬����һ������Z��X������������� | |
| D�� | ��������ѹǿ���䣬����һ�����Ķ������壬Y��Ũ�Ȳ��� |
| A�� | ������H2������Ϊ0.02g | |
| B�� | ���������ԭ����Ϊ1.204��1023�� | |
| C�� | ������O2�����Ϊ0.224 L | |
| D�� | ����H2���������ϵ���0.02mol Zn���������ᷴӦ����H2���� |
| A�� | KNO3 | B�� | CH3COONa | C�� | CuSO4 | D�� | Na2CO3 |
| A�� | ����մ�����ϣ�Ӧ������ŨNaOH��Һ��ϴ | |
| B�� | �ƾ����������㵹ʧ��ʱ��������ʪ������ | |
| C�� | ��ȼCO��CH4�ȿ�ȼ������ǰ�������������Ĵ��� | |
| D�� | ���Թ����Һ�����ʱ��Һ�����һ�㲻�����Թ��ݻ���$\frac{1}{3}$ |
| ���� | Na+ | Al3+ | Cl- | X |
| ���ʵ��� | 2a | a | a | b |
| A�� | NO3-��2a | B�� | CO32-��2a | C�� | OH-��4a | D�� | SO42-��2a |
| A�� | ��̼������Һ����������������2CO32-+SO2+H2O=2HCO3-+HSO3- | |
| B�� | ��������Һ��ͨ������������̼��SiO32-+2CO2+3H2O�TH4SiO4��+2HCO3- | |
| C�� | �����������Һ�м���Ba��OH��2��Һ���պó�����ȫ��2H++SO42-+Ba2++2OH-=2H2O+BaSO4�� | |
| D�� | С�մ���Һ�м�������ij���ʯ��ˮ��2HCO3-+Ca2++2OH-�TCaCO3��+2H2O+CO32- |
| A�� | c��Fe3+��=0.1mol/L����Һ�У�K+��Mg2+��SCN-��SO42- | |
| B�� | c��H+��=1mol/L����Һ�У�Cu2+��Na+��ClO-��S2- | |
| C�� | ��ʹ��̪������Һ�У�Na+��Ba2+��NO3-��Cl- | |
| D�� | pH=2����Һ�У�Na+��Ag+��I-��NO3- |
 ij�Ͼ����Ͽɲ������з����������������ϸ���������ǿ�ȣ�ʹ�������õ����ʣ�ʵ��װ����ͼ�����Ⱦ۱�ϩ�����ϵõ��IJ��������
ij�Ͼ����Ͽɲ������з����������������ϸ���������ǿ�ȣ�ʹ�������õ����ʣ�ʵ��װ����ͼ�����Ⱦ۱�ϩ�����ϵõ��IJ��������| ���� | ���� | ���� | ��ϩ | ��ϩ | �� | �ױ� | ̼ |
| ����������%�� | 12 | 24 | 12 | 16 | 20 | 10 | 6 |
A�в�����$\stackrel{��}{��}$��ʯ$\stackrel{��}{��}$ ��Ȳ$\stackrel{��}{��}$����Ȳ
д����Ӧ�ڢ۵Ļ�ѧ����ʽ��CaC2+2H2O=Ca��OH��2+C2H2������nC2H2$\stackrel{һ������}{��}$
��2���Թ�B�ռ��õIJ�Ʒ�У�����ʹ���Ը��������Һ��ɫ�����ʣ���һ�Ȼ�����4�֣���д�������������ᷴӦ�Ļ�ѧ����ʽ
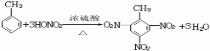 ��
����3����ƿC�й۲쵽�������ػ�ɫ��Һ����ɫ����ɫ������ˮ������գ�ʣ�����徭������ƽ����Է�������Ϊ4.8
��4��ij�л��ﳣ����ΪҺ�壬����ϩ��Ϊͬϵ���Է���������86���ڣ�����HBr�ӳɲ���ֻ��һ�ֽṹ�����л���Ľṹ��ʽ����ΪCH3CH2CH=CHCH2CH3����CH3��2C=C��CH3��2��