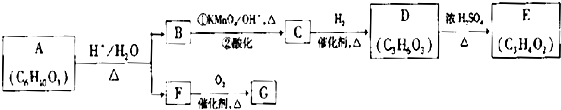
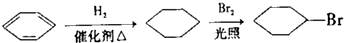
��Ŀ����
�ۻ�ѧ��ѡ���л���ѧ�����ݣ�15�֣���
��ŵ������������Ľ�ʹ��ѡ��ҩ����ŵ຺ϳ�·�����£�

��1����ŵ��к��������ţ�����������-NHCO-���⣬�����ֹ��������ƣ��ߣߣߣߡ��ߣߣߣߡ�
��2��д��������������A��ͬ���칹�����Ŀ�ߣߣߣߡ�
��A����FeCl3��Һ������ɫ��Ӧ����1molA����2molNaOH��Ӧ��
��3��C��D�У���������NaOH�������ǣ��ߣߣߣߣߣߣߣߣߣߣߣߡ�
��4������ŵ຺ϳ�·�ߵõ���ʾ�����ü���ױ���ClCH2COCl����C2H5��2NH�����Լ����ã��ϳ�������������ں����ϲ��䷴Ӧ��ڷ����ڲ��������
��֪��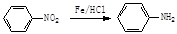
��15�֣�����1���ǻ����Ѽ���2�֣��� ��2��6 ��2�֣�
��3����ȡ������HCl���ã�ʹƽ��������D�ķ����ƶ������D�IJ��ʣ�2�֣���
��4��
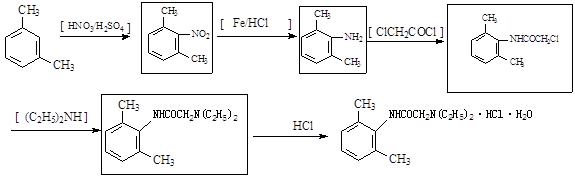
����:

��ϰ��ϵ�д�
�����Ŀ






 ��R1��R2��R3����������
��R1��R2��R3����������




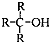 ������������ȩ��ͪ��
������������ȩ��ͪ��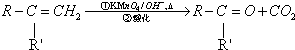 ��R��R��ɱ�ʾ����������ţ�
��R��R��ɱ�ʾ����������ţ�