��Ŀ����
2�� A��B��C��D��E����Ԫ�����ڱ���ǰ20��Ԫ�أ���ԭ��������������B��C��Dͬ���ڣ�A��Dͬ���壬E������Ԫ�ؼȲ���ͬ����Ҳ����ͬ���壮B��C��D������������Ӧˮ������ܻ������Ӧ�����κ�ˮ��A��E���γ����ӻ�����侧���ṹ��ͼ��ʾ��
A��B��C��D��E����Ԫ�����ڱ���ǰ20��Ԫ�أ���ԭ��������������B��C��Dͬ���ڣ�A��Dͬ���壬E������Ԫ�ؼȲ���ͬ����Ҳ����ͬ���壮B��C��D������������Ӧˮ������ܻ������Ӧ�����κ�ˮ��A��E���γ����ӻ�����侧���ṹ��ͼ��ʾ����1��A��E���γɻ�����ĵ���ʽ��
 ��
����2��A��B��C��D�ĸ�Ԫ�ص�ԭ�Ӱ뾶��С�����˳��ΪNa��Al��Cl��F����Ԫ�ط��ű�ʾ����
��3��A��E�γɵĻ�����ľ����У�ÿ����������Χ��������Ҿ�����ȵ������ӹ���12����
��4����֪A��E���γɻ����ᄃ��ľ������Ϊ1.6��10-22 cm3����A��E��ɵ����ӻ�������ܶ�Ϊ���������һλС����3.3g/cm3��
���� A��B��C��D��E����Ԫ�����ڱ��е�ǰ20��Ԫ�أ�ԭ��������������B��C��D������������ˮ����������Ͼ��ܷ�����Ӧ�����κ�ˮ��Ӧ������������ǿ�ǿ�ᷴӦ������֪BΪNa��CΪAl��E������Ԫ�ؼȲ���ͬ����Ҳ����ͬ���壬ֻ�ܴ��ڵ������ڢ�A�壬��EΪCa��A��E���γ����ӻ�����侧���ṹ��ͼ��ʾ������������������������Ŀ֮��Ϊ1��4��$\frac{1}{8}$=2��1���������ӱ���-1�ۣ���A��Dͬ���壬��AΪFԪ�ء�DΪClԪ�أ��ݴ˽��
��� �⣺A��B��C��D��E����Ԫ�����ڱ��е�ǰ20��Ԫ�أ�ԭ��������������B��C��D������������ˮ����������Ͼ��ܷ�����Ӧ�����κ�ˮ��Ӧ������������ǿ�ǿ�ᷴӦ������֪BΪNa��CΪAl��E������Ԫ�ؼȲ���ͬ����Ҳ����ͬ���壬ֻ�ܴ��ڵ������ڢ�A�壬��EΪCa��A��E���γ����ӻ�����侧���ṹ��ͼ��ʾ������������������������Ŀ֮��Ϊ1��4��$\frac{1}{8}$=2��1���������ӱ���-1�ۣ���A��Dͬ���壬��AΪFԪ�ء�DΪClԪ�أ�
��1��A��E���γɻ�����ΪCaF2������ʽ�� ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
��2��ͬ�����������ԭ�Ӱ뾶��С��ͬ�������϶���ԭ�Ӱ뾶����ԭ�Ӱ뾶��Na��Al��Cl��F���ʴ�Ϊ��Na��Al��Cl��F��
��3���Զ���������Ϊ�о�������������Ҿ�����ȵ�������λ�����ģ�ÿ������Ϊ8���������ã�ÿ������Ϊ2���������ã��ʹ���$\frac{3��8}{2}$=12�����ʴ�Ϊ��12��
��4�������з���������ĿΪ8����������ĿΪ4���ʾ�������Ϊ4��$\frac{78}{{N}_{A}}$g���������Ϊ1.6��10-22 cm3����A��E��ɵ����ӻ�������ܶ�Ϊ4��$\frac{78g}{6.02��1{0}^{23}}$g��1.6��10-22 cm3=3.3g/cm3��
�ʴ�Ϊ��3.3g/cm3��
���� �����Ƕ����ʽṹ�Ŀ��飬�漰����ʽ��ԭ�Ӱ뾶�Ƚϡ������йؼ���ȣ�ע�����þ�̯�����㾧���йؼ��㣮

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | ʳ����ڱ����л�������ʵ����� | |
| B�� | ʵ�����Ʊ�CO2ʱ����ͨ������ʯ��ʯ���������ӿ췴Ӧ���� | |
| C�� | ��ȡ��������ʱ����Ũ�����������ɼӿ�������Ӧ������ | |
| D�� | ʵ������ȡH2ʱ������п������п���Խ��ͷ�Ӧ������ |
������ 0.50mol/L NaOH��Һ
��1����ʵ���д�ԼҪʹ��245mL NaOH��Һ��������Һ��Ҫ��250ml����ƿ����������Ҫ����NaOH����5.0g��
��2������ͼ��ѡ�����NaOH��������Ҫ������������ţ�a b e��
| ���� | ������ƽ�������룩 | С�ձ� | ����ǯ | ������ | ҩ�� | ��Ͳ |
| ���� |  |  |  |  |  |  |
| ��� | a | b | c | d | e | f |
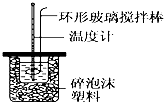
��1��д��ϡ�����ϡ����������Һ��Ӧ��ʾ�к��ȵ��Ȼ�ѧ����ʽ���к�����ֵΪ57.3kJ/mol����NaOH��aq��+$\frac{1}{2}$H2SO4��aq���T$\frac{1}{2}$Na2SO4��aq��+H2O��l����H=-57.3 kJ/mol��
��2��ȡ50mL NaOH��Һ��30mL�������ʵ�飬ʵ�����������
������д�±���ƽ���¶Ȳ�Ŀո�
| �¶� ʵ����� | ��ʼ�¶�t1/�� | ��ֹ�� ��t2/�� | ƽ���¶Ȳ� ��t2-t1��/�� | ||
| H2SO4��Һ | NaOH��Һ | ƽ��ֵ | |||
| 1 | 26.2 | 26.0 | 26.1 | 30.1 | �����4.0�� �� |
| 2 | 27.0 | 27.4 | 27.2 | 33.3 | |
| 3 | 25.9 | 25.9 | 25.9 | 29.8 | |
| 4 | 26.4 | 26.2 | 26.3 | 30.4 | |
c=4.18J/��g•�棩�����к��ȡ�H=-53.5kJ/mol������С�����һλ����
������ʵ��������ֵ��57.3kJ/mol��ƫ�����ƫ���ԭ������ǣ�����ţ�acd��
a��ʵ��װ�ñ��¡�����Ч����
b������ȡNaOH��Һ�����ʱ���Ӷ���
c���ֶ�ΰ�NaOH��Һ����ʢ��ϡ�����С�ձ���
d�����¶ȼƲⶨNaOH��Һ��ʼ�¶Ⱥ�ֱ�ӲⶨH2SO4��Һ���¶�
����֪ǿ����ǿ����ϡ��Һ�ﷴӦ���к��ȿɱ�ʾΪ
H+��aq��+OH-��aq���TH2O��l����H=-57.3kJ•mol-1�����з�Ӧ��
CH3COOH��aq��+NaOH��aq���TCH3COONa��aq��+H2O��l����H=-Q1 kJ•mol-1
$\frac{1}{2}$H2SO4��Ũ��+NaOH��aq���T$\frac{1}{2}$Na2SO4��aq��+H2O��l����H=-Q2 kJ•mol-1
HNO3��aq��+NaOH��aq���TNaNO3��aq��+H2O��l����H=-Q3 kJ•mol-1
������Ӧ������Һ�н��У�������Q1��Q2��Q3�Ĺ�ϵ��ȷ����A
A��Q2��Q3��Q1 B��Q2��Q1��Q3 C��Q1=Q2=Q3 D��Q2=Q3��Q1��
| A�� | ú��ʯ�͵Ȼ�ʯȼ��ȼ�չ������ŷŵķ������γ��������Ҫԭ������ָ����PHֵС��7����ˮ | |
| B�� | ú��ʯ�Ͷ�����һ����Դ����Ȼ�����ڶ�����Դ | |
| C�� | ��ú������������ʯ��ɼ���ȼ�չ����к�������ŷŴӶ�������Ⱦ | |
| D�� | ��������������ϸСҺ���������ɢ���������γɵ�һ�����ܽ� |
| A�� | x��y���ڵ������ļ��ҵķ��Ӹ���֮�� | |
| B�� | x��y����ͬ��ͬѹ�¼��ҵ��ܶ�֮�� | |
| C�� | x��y���ڼ����ҵ���Է�������֮�� | |
| D�� | x��y����ͬ��ͬ����µ������ļ��ҵ�ѹǿ֮�� |
| A�� | ԭ�Ӻ�������Ų�ʽΪ1s2��Xԭ����ԭ�Ӻ�������Ų�ʽΪ1s22s2��Yԭ�� | |
| B�� | ԭ�Ӻ���M���Ͻ����������ӵ�Xԭ����ԭ�Ӻ���N���Ͻ��������ӵ�Yԭ�� | |
| C�� | 2p�����ֻ��һ���չ����Xԭ����3p�����ֻ��һ���չ����Yԭ�� | |
| D�� | ����㶼ֻ��һ�����ӵ�X��Yԭ�� |
| A�� | ��H1����H2����H3 | B�� | ��H2����H3����H1 | C�� | ��H1=��H2=��H3 | D�� | ��H2����H3����H1 |
| A�� | ũҩ�ɺ����л��չ��Ϊ�������Խϵ͵ĺ����л���� | |
| B�� | �����ڱ��н�����ǽ����ķֽ紦�������ҵ��뵼����ϣ���衢��� | |
| C�� | �����ڹ���Ԫ����Ѱ�Ҵ��������¡���ʴ�ĺϽ���� | |
| D�� | �������ڱ�������Ԫ���������ƵĹ��ɣ������ڱ�һ��������Ѱ��Ԫ�أ��������ʵ�����;����Ϊ�൱��Ч�ķ��� |